Ibrexafungerp Versus Placebo for Vulvovaginal Candidiasis Treatment: A Phase 3, Randomized, Controlled Superiority Trial (VANISH 303)
- PMID: 34467969
- PMCID: PMC9187327
- DOI: 10.1093/cid/ciab750
Ibrexafungerp Versus Placebo for Vulvovaginal Candidiasis Treatment: A Phase 3, Randomized, Controlled Superiority Trial (VANISH 303)
Abstract
Background: Current treatment of vulvovaginal candidiasis (VVC) is largely limited to azole therapy. Ibrexafungerp is a first-in-class triterpenoid antifungal with broad-spectrum anti-Candida fungicidal activity. The objective of this study was to evaluate the efficacy and safety of ibrexafungerp compared with placebo in patients with acute VVC.
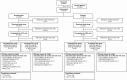
Methods: Patients were randomly assigned 2:1 to receive ibrexafungerp (300 mg twice for 1 day) or placebo. The primary endpoint was the percentage of patients with a clinical cure (complete resolution of vulvovaginal signs and symptoms [VSS] = 0) at test-of-cure (day 11 ± 3). Secondary endpoints included the percentage of patients with mycological eradication, overall success (clinical cure and mycological eradication), clinical improvement (VSS ≤ 1) at test-of-cure, and symptom resolution at follow-up (day 25 ± 4).
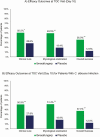
Results: Patients receiving ibrexafungerp had significantly higher rates of clinical cure (50.5% [95/188] vs 28.6% [28/98]; P = .001), mycological eradication (49.5% [93/188] vs 19.4% [19/98]; P < .001), and overall success (36.0% [64/178] vs 12.6% [12/95]; P < .001) compared with placebo. Symptom resolution was sustained and further increased with ibrexafungerp compared with placebo (59.6% [112/188] vs 44.9% [44/98]; P = .009) at follow-up. Post hoc analysis showed similar rates of clinical cure and clinical improvement at test-of-cure for Black patients (54.8% [40/73] and 63.4% [47/73], respectively) and patients with a body mass index >35 (54.5% [24/44] and 68.2% [30/44], respectively) compared with overall rates. Ibrexafungerp was well tolerated. Adverse events were primarily gastrointestinal and mild in severity.
Conclusions: Ibrexafungerp provides a promising safe and efficacious oral treatment that mechanistically differs from current azole treatment options for acute VVC.
Keywords: Candida albicans; SCY-078; ibrexafungerp; placebo; vulvovaginal candidiasis.
© The Author(s) 2021. Published by Oxford University Press for the Infectious Diseases Society of America.
Figures
Comment in
-
Ibrexafungerp to treat acute vulvovaginal candidiasis.Nat Rev Urol. 2021 Nov;18(11):638. doi: 10.1038/s41585-021-00522-9. Nat Rev Urol. 2021. PMID: 34548655 No abstract available.
Similar articles
-
Efficacy and safety of oral ibrexafungerp for the treatment of acute vulvovaginal candidiasis: a global phase 3, randomised, placebo-controlled superiority study (VANISH 306).BJOG. 2022 Feb;129(3):412-420. doi: 10.1111/1471-0528.16972. Epub 2021 Nov 8. BJOG. 2022. PMID: 34676663 Free PMC article. Clinical Trial.
-
Efficacy and safety of oral ibrexafungerp in Chinese patients with vulvovaginal candidiasis: a phase III, randomized, double-blind study.Infection. 2024 Oct;52(5):1787-1797. doi: 10.1007/s15010-024-02233-w. Epub 2024 Apr 3. Infection. 2024. PMID: 38568411 Free PMC article. Clinical Trial.
-
Oral Ibrexafungerp for Vulvovaginal Candidiasis Treatment: An Analysis of VANISH 303 and VANISH 306.J Womens Health (Larchmt). 2023 Feb;32(2):178-186. doi: 10.1089/jwh.2022.0132. Epub 2022 Oct 17. J Womens Health (Larchmt). 2023. PMID: 36255448 Free PMC article. Clinical Trial.
-
Ibrexafungerp for the Treatment of Vulvovaginal Candidiasis: Design, Development and Place in Therapy.Drug Des Devel Ther. 2023 Feb 7;17:363-367. doi: 10.2147/DDDT.S339349. eCollection 2023. Drug Des Devel Ther. 2023. PMID: 36785761 Free PMC article. Review.
-
Ibrexafungerp in the Treatment of Vulvovaginal Candidiasis.Ann Pharmacother. 2023 Jan;57(1):99-106. doi: 10.1177/10600280221091301. Epub 2022 May 2. Ann Pharmacother. 2023. PMID: 35502451 Review.
Cited by
-
Development and characterization of amphotericin B nanoemulsion-loaded mucoadhesive gel for treatment of vulvovaginal candidiasis.Heliyon. 2022 Nov 12;8(11):e11489. doi: 10.1016/j.heliyon.2022.e11489. eCollection 2022 Nov. Heliyon. 2022. PMID: 36411885 Free PMC article.
-
In Vitro Antifungal Activity of Ibrexafungerp (SCY-078) Against Contemporary Blood Isolates From Medically Relevant Species of Candida: A European Study.Front Cell Infect Microbiol. 2022 May 16;12:906563. doi: 10.3389/fcimb.2022.906563. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35651755 Free PMC article.
-
Efficacy and safety of ibrexafungerp in the treatment of vulvovaginal candidiasis: A meta-analysis of randomized controlled trials.Heliyon. 2024 Apr 5;10(8):e28776. doi: 10.1016/j.heliyon.2024.e28776. eCollection 2024 Apr 30. Heliyon. 2024. PMID: 38628772 Free PMC article.
-
Fungal infections: Pathogenesis, antifungals and alternate treatment approaches.Curr Res Microb Sci. 2022 Apr 27;3:100137. doi: 10.1016/j.crmicr.2022.100137. eCollection 2022. Curr Res Microb Sci. 2022. PMID: 35909631 Free PMC article. Review.
-
Treatment of Vulvovaginal Candidiasis-An Overview of Guidelines and the Latest Treatment Methods.J Clin Med. 2023 Aug 18;12(16):5376. doi: 10.3390/jcm12165376. J Clin Med. 2023. PMID: 37629418 Free PMC article. Review.
References
-
- Mendling W, Brasch J, Cornely OA, et al. . Guideline: vulvovaginal candidosis (AWMF 015/072), S2k (excluding chronic mucocutaneous candidosis). Mycoses 2015; 58:1–15. - PubMed
-
- Jeanmonod R, Jeanmonod D. Vaginal candidiasis. Available at: https://www.ncbi.nlm.nih.gov/books/. Accessed 9 March 2021.
-
- Sobel JD. Recurrent vulvovaginal candidiasis. Am J Obstet Gynecol 2016; 214:15–21. - PubMed

