A therapeutic approach to pantothenate kinase associated neurodegeneration: a pilot study
- PMID: 39609877
- PMCID: PMC11606047
- DOI: 10.1186/s13023-024-03453-x
A therapeutic approach to pantothenate kinase associated neurodegeneration: a pilot study
Abstract
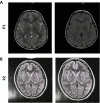
Background: Neurodegeneration with brain iron accumulation (NBIA) is a group of genetic neurological disorders frequently associated with iron accumulation in the basal nuclei of the brain characterized by progressive spasticity, dystonia, muscle rigidity, neuropsychiatric symptoms, and retinal degeneration or optic nerve atrophy. Pantothenate kinase-associated neurodegeneration (PKAN) is one of the most widespread NBIA disorders. The diagnosis of PKAN is established with clinical features and the "eye of the tiger" sign identified on brain MRI and the identification of biallelic pantothenate kinase 2 (PANK2) pathogenic variants on molecular genetic testing. PANK2 catalyzes the first reaction of coenzyme A (CoA) biosynthesis, thus, altered PANK2 activity is expected to induce CoA deficiency as well as low levels of essential metabolic intermediates such as 4'-phosphopantetheine which is a necessary cofactor for critical proteins involved in cytosolic and mitochondrial pathways such as fatty acid biosynthesis, mitochondrial respiratory complex I assembly and lysine and tetrahydrofolate metabolism, among other metabolic processes.
Methods: In this manuscript, we examined the effect of a multitarget complex supplements (pantothenate, pantethine, omega-3 and vitamin E) on in vitro patient-derived cellular models and the clinical outcome of the adjuvant supplements in combination with the baseline neurological medication in three PKAN patients.
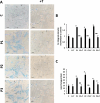
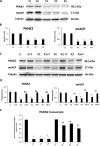
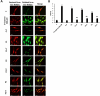
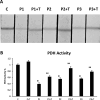
Results: Multitarget complex supplements significantly reduced iron accumulation and increased PANK2 and ACP expression levels in the cellular models derived from all three PKAN patients. In addition, the adjunct treatment to the standard neurological medication improved or stabilized the clinical symptoms of patients.
Conclusions: Our results suggest that multitarget complex supplements can be clinically useful as augmentation therapy for PKAN patients harboring pathogenic variants with residual enzyme levels.
Trial registration: CAAE: 58219522.6.0000.5330. Registered 25 May 2022-Retrospectively registered, https://plataformabrasil.saude.gov.br/visao/pesquisador/gerirPesquisa/gerirPesquisaAgrupador.jsf .
© 2024. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The study was approved by The Ethical Committee of Hospital Universitario Virgen del Rocío and Virgen Macarena of Seville, protocol code BRAINCURE16, following the Spanish laws, the principles of the Declaration of Helsinki, and the Guideline for Good Clinical Practices; and by the Ethics Committee of Moinhos de Vento Hospital (CAAE: 58219522.6.0000.5330) and all the parents or legal guardians of subjects signed the written informed consent forms before any procedure was done. Consent for publication: Consent for publication were obtained from all parents or legal guardians. Competing interests: The authors declare that they have no competing interests.
Figures

Similar articles
-
Depressing time: Waiting, melancholia, and the psychoanalytic practice of care.In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. PMID: 36137063 Free Books & Documents. Review.
-
A Potential Citrate Shunt in Erythrocytes of PKAN Patients Caused by Mutations in Pantothenate Kinase 2.Biomolecules. 2022 Feb 18;12(2):325. doi: 10.3390/biom12020325. Biomolecules. 2022. PMID: 35204826 Free PMC article.
-
Antioxidants for female subfertility.Cochrane Database Syst Rev. 2020 Aug 27;8(8):CD007807. doi: 10.1002/14651858.CD007807.pub4. Cochrane Database Syst Rev. 2020. PMID: 32851663 Free PMC article.
-
Qualitative evidence synthesis informing our understanding of people's perceptions and experiences of targeted digital communication.Cochrane Database Syst Rev. 2019 Oct 23;10(10):ED000141. doi: 10.1002/14651858.ED000141. Cochrane Database Syst Rev. 2019. PMID: 31643081 Free PMC article.
-
Antioxidants for female subfertility.Cochrane Database Syst Rev. 2017 Jul 28;7(7):CD007807. doi: 10.1002/14651858.CD007807.pub3. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Aug 27;8:CD007807. doi: 10.1002/14651858.CD007807.pub4. PMID: 28752910 Free PMC article. Updated. Review.
References
-
- Zorzi G, et al. Iron-related MRI images in patients with pantothenate kinase-associated neurodegeneration (PKAN) treated with deferiprone: results of a phase II pilot trial. Mov Disord. 2011;26(9):1756–9. - PubMed
-
- Hayflick SJ. Unraveling the Hallervorden-Spatz syndrome: pantothenate kinase-associated neurodegeneration is the name. Curr Opin Pediatr. 2003;15(6):572–7. - PubMed
-
- Hayflick SJ. Neurodegeneration with brain iron accumulation: from genes to pathogenesis. Semin Pediatr Neurol. 2006;13(3):182–5. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

