Elafibranor alleviates alcohol-related liver fibrosis by restoring intestinal barrier function
- PMID: 39575408
- PMCID: PMC11572637
- DOI: 10.3748/wjg.v30.i43.4660
Elafibranor alleviates alcohol-related liver fibrosis by restoring intestinal barrier function
Abstract
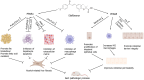
We discuss the article by Koizumi et al published in the World Journal of Gastroenterology. Our focus is on the therapeutic targets for fibrosis associated with alcohol-related liver disease (ALD) and the mechanism of action of elafibranor (EFN), a dual agonist of peroxisome proliferator-activated receptor α (PPARα) and peroxisome PPAR δ (PPARδ). EFN is currently in phase III clinical trials for the treatment of metabolic dysfunction-associated fatty liver disease and primary biliary cholangitis. ALD progresses from alcoholic fatty liver to alcoholic steatohepatitis (ASH), with chronic ASH eventually leading to fibrosis, cirrhosis, and, in some cases, hepatocellular carcinoma. The pathogenesis of ALD is driven by hepatic steatosis, oxidative stress, and acetaldehyde toxicity. Alcohol consumption disrupts lipid metabolism by inactivating PPARα, exacerbating the progression of ALD. EFN primarily activates PPARα, promoting lipolysis and β-oxidation in ethanol-stimulated HepG2 cells, which significantly reduces hepatic steatosis, apoptosis, and fibrosis in an ALD mouse model. Additionally, alcohol disrupts the gut-liver axis at several interconnected levels, contributing to a proinflammatory environment in the liver. EFN helps alleviate intestinal hyperpermeability by restoring tight junction protein expression and autophagy, inhibiting apoptosis and inflammatory responses, and enhancing intestinal barrier function through PPARδ activation.
Keywords: Apoptosis; Autophagy; Ethanol; Gut barrier function; Liver fibrosis; Peroxisome proliferator-activated receptor.
©The Author(s) 2024. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: The authors declare that they have no conflict of interest to disclose.
Figures
Similar articles
-
Effects of elafibranor on liver fibrosis and gut barrier function in a mouse model of alcohol-associated liver disease.World J Gastroenterol. 2024 Jul 28;30(28):3428-3446. doi: 10.3748/wjg.v30.i28.3428. World J Gastroenterol. 2024. PMID: 39091710 Free PMC article.
-
Elafibranor: A promising therapeutic approach for liver fibrosis and gut barrier dysfunction in alcohol-associated liver disease.World J Gastroenterol. 2025 Jan 21;31(3):98783. doi: 10.3748/wjg.v31.i3.98783. World J Gastroenterol. 2025. PMID: 39839906 Free PMC article. Review.
-
Elafibranor interrupts adipose dysfunction-mediated gut and liver injury in mice with alcoholic steatohepatitis.Clin Sci (Lond). 2019 Feb 13;133(3):531-544. doi: 10.1042/CS20180873. Print 2019 Feb 14. Clin Sci (Lond). 2019. PMID: 30602573
-
Dual peroxisome proliferator-activated receptor α/δ agonists: Hope for the treatment of alcohol-associated liver disease?World J Gastroenterol. 2024 Oct 7;30(37):4163-4167. doi: 10.3748/wjg.v30.i37.4163. World J Gastroenterol. 2024. PMID: 39474402 Free PMC article.
-
Alcoholic fatty liver: its pathogenesis and mechanism of progression to inflammation and fibrosis.Alcohol. 2004 Aug;34(1):9-19. doi: 10.1016/j.alcohol.2004.07.008. Alcohol. 2004. PMID: 15670660 Review.
References
-
- Caputo F, Lungaro L, Guarino M, Costanzini A, Caio G, Testino G, DE Giorgio R. Alcohol-related diseases: from metabolism to the main effect on the body. Minerva Med. 2024 - PubMed
-
- Seitz HK, Bataller R, Cortez-Pinto H, Gao B, Gual A, Lackner C, Mathurin P, Mueller S, Szabo G, Tsukamoto H. Alcoholic liver disease. Nat Rev Dis Primers. 2018;4:16. - PubMed
-
- Seitz HK, Stickel F. Molecular mechanisms of alcohol-mediated carcinogenesis. Nat Rev Cancer. 2007;7:599–612. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

