Drivers and consequences of microbial community coalescence
- PMID: 39288091
- PMCID: PMC11447283
- DOI: 10.1093/ismejo/wrae179
Drivers and consequences of microbial community coalescence
Abstract
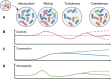
Microbial communities are undergoing unprecedented dispersion and amalgamation across diverse ecosystems, thereby exerting profound and pervasive influences on microbial assemblages and ecosystem dynamics. This review delves into the phenomenon of community coalescence, offering an ecological overview that outlines its four-step process and elucidates the intrinsic interconnections in the context of community assembly. We examine pivotal mechanisms driving community coalescence, with a particular emphasis on elucidating the fates of both source and resident microbial communities and the consequential impacts on the ecosystem. Finally, we proffer recommendations to guide researchers in this rapidly evolving domain, facilitating deeper insights into the ecological ramifications of microbial community coalescence.
Keywords: community stability; dispersal; disturbance; interspecies interaction; invasion; microbial community dynamic; microbial inoculation.
© The Author(s) 2024. Published by Oxford University Press on behalf of the International Society for Microbial Ecology.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Linking microbial community coalescence to ecological diversity, community assembly and species coexistence in a typical subhumid river catchment in northern China.Sci Total Environ. 2024 Aug 15;938:173367. doi: 10.1016/j.scitotenv.2024.173367. Epub 2024 May 23. Sci Total Environ. 2024. PMID: 38796011
-
Assembly process and co-occurrence network of microbial community in response to free ammonia gradient distribution.Microbiol Spectr. 2024 Sep 3;12(9):e0105124. doi: 10.1128/spectrum.01051-24. Epub 2024 Jul 26. Microbiol Spectr. 2024. PMID: 39058029 Free PMC article.
-
Dynamics of the sedimentary bacterial communities in a plain river network: similar coalescence patterns with bacterioplankton communities driven by distinct assembly processes.Appl Environ Microbiol. 2023 Dec 21;89(12):e0146523. doi: 10.1128/aem.01465-23. Epub 2023 Nov 21. Appl Environ Microbiol. 2023. PMID: 38092675 Free PMC article.
-
Application of the microbial community coalescence concept to riverine networks.Biol Rev Camb Philos Soc. 2018 Nov;93(4):1832-1845. doi: 10.1111/brv.12422. Epub 2018 Apr 26. Biol Rev Camb Philos Soc. 2018. PMID: 29700966 Review.
-
A conceptual framework for invasion in microbial communities.ISME J. 2016 Dec;10(12):2773-2775. doi: 10.1038/ismej.2016.75. Epub 2016 May 3. ISME J. 2016. PMID: 27137125 Free PMC article. Review.
References
-
- Rillig MC, Lehmann A, Aguilar-Trigueros CA et al. Soil microbes and community coalescence. Pedobiologia 2016;59:37–40. 10.1016/j.pedobi.2016.01.001 - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

