High-quality physiology of Alcanivorax borkumensis SK2 producing glycolipids enables efficient stirred-tank bioreactor cultivation
- PMID: 38084272
- PMCID: PMC10710537
- DOI: 10.3389/fbioe.2023.1325019
High-quality physiology of Alcanivorax borkumensis SK2 producing glycolipids enables efficient stirred-tank bioreactor cultivation
Abstract
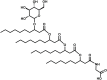
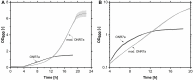
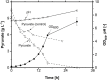
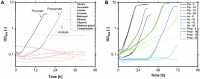
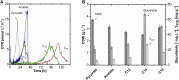
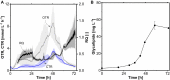
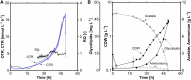
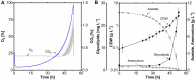
Glycine-glucolipid, a glycolipid, is natively synthesized by the marine bacterium Alcanivorax borkumensis SK2. A. borkumensis is a Gram-negative, non-motile, aerobic, halophilic, rod-shaped γ-proteobacterium, classified as an obligate hydrocarbonoclastic bacterium. Naturally, this bacterium exists in low cell numbers in unpolluted marine environments, but during oil spills, the cell number significantly increases and can account for up to 90% of the microbial community responsible for oil degradation. This growth surge is attributed to two remarkable abilities: hydrocarbon degradation and membrane-associated biosurfactant production. This study aimed to characterize and enhance the growth and biosurfactant production of A. borkumensis, which initially exhibited poor growth in the previously published ONR7a, a defined salt medium. Various online analytic tools for monitoring growth were employed to optimize the published medium, leading to improved growth rates and elongated growth on pyruvate as a carbon source. The modified medium was supplemented with different carbon sources to stimulate glycine-glucolipid production. Pyruvate, acetate, and various hydrophobic carbon sources were utilized for glycolipid production. Growth was monitored via online determined oxygen transfer rate in shake flasks, while a recently published hyphenated HPLC-MS method was used for glycine-glucolipid analytics. To transfer into 3 L stirred-tank bioreactor, aerated batch fermentations were conducted using n-tetradecane and acetate as carbon sources. The challenge of foam formation was overcome using bubble-free membrane aeration with acetate as the carbon source. In conclusion, the growth kinetics of A. borkumensis and glycine-glucolipid production were significantly improved, while reaching product titers relevant for applications remains a challenge.
Keywords: acetate; alkanes; bioremediation; biosurfactant; glycolipid; hydrocarbonoclastic bacteria; hydrocarbons; membrane aeration.
Copyright © 2023 Karmainski, Dielentheis-Frenken, Lipa, Phan, Blank and Tiso.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures

Similar articles
-
Identification and quantification of biosurfactants produced by the marine bacterium Alcanivorax borkumensis by hyphenated techniques.Anal Bioanal Chem. 2023 Dec;415(29-30):7067-7084. doi: 10.1007/s00216-023-04972-5. Epub 2023 Oct 11. Anal Bioanal Chem. 2023. PMID: 37819435 Free PMC article.
-
The Glycine-Glucolipid of Alcanivorax borkumensis Is Resident to the Bacterial Cell Wall.Appl Environ Microbiol. 2022 Aug 23;88(16):e0112622. doi: 10.1128/aem.01126-22. Epub 2022 Aug 8. Appl Environ Microbiol. 2022. PMID: 35938787 Free PMC article.
-
Membrane Fatty Acid Composition and Cell Surface Hydrophobicity of Marine Hydrocarbonoclastic Alcanivorax borkumensis SK2 Grown on Diesel, Biodiesel and Rapeseed Oil as Carbon Sources.Molecules. 2018 Jun 13;23(6):1432. doi: 10.3390/molecules23061432. Molecules. 2018. PMID: 29899233 Free PMC article.
-
Genome sequence completed of Alcanivorax borkumensis, a hydrocarbon-degrading bacterium that plays a global role in oil removal from marine systems.J Biotechnol. 2003 Dec 19;106(2-3):215-20. doi: 10.1016/j.jbiotec.2003.07.013. J Biotechnol. 2003. PMID: 14651863 Review.
-
Occurrence, production, and export of lipophilic compounds by hydrocarbonoclastic marine bacteria and their potential use to produce bulk chemicals from hydrocarbons.Appl Microbiol Biotechnol. 2010 May;86(6):1693-706. doi: 10.1007/s00253-010-2515-5. Epub 2010 Mar 31. Appl Microbiol Biotechnol. 2010. PMID: 20354694 Review.
Cited by
-
Unlocking the potential of biosurfactants: Innovations in metabolic and genetic engineering for sustainable industrial and environmental solutions.Biotechnol Notes. 2024 Jul 25;5:111-119. doi: 10.1016/j.biotno.2024.07.001. eCollection 2024. Biotechnol Notes. 2024. PMID: 39416688 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

