Innate Immune System in the Context of Radiation Therapy for Cancer
- PMID: 37568788
- PMCID: PMC10417569
- DOI: 10.3390/cancers15153972
Innate Immune System in the Context of Radiation Therapy for Cancer
Abstract
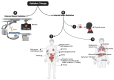
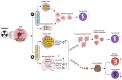
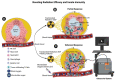
Radiation therapy (RT) remains an integral component of modern oncology care, with most cancer patients receiving radiation as a part of their treatment plan. The main goal of ionizing RT is to control the local tumor burden by inducing DNA damage and apoptosis within the tumor cells. The advancement in RT, including intensity-modulated RT (IMRT), stereotactic body RT (SBRT), image-guided RT, and proton therapy, have increased the efficacy of RT, equipping clinicians with techniques to ensure precise and safe administration of radiation doses to tumor cells. In this review, we present the technological advancement in various types of RT methods and highlight their clinical utility and associated limitations. This review provides insights into how RT modulates innate immune signaling and the key players involved in modulating innate immune responses, which have not been well documented earlier. Apoptosis of cancer cells following RT triggers immune systems that contribute to the eradication of tumors through innate and adoptive immunity. The innate immune system consists of various cell types, including macrophages, dendritic cells, and natural killer cells, which serve as key mediators of innate immunity in response to RT. This review will concentrate on the significance of the innate myeloid and lymphoid lineages in anti-tumorigenic processes triggered by RT. Furthermore, we will explore essential strategies to enhance RT efficacy. This review can serve as a platform for researchers to comprehend the clinical application and limitations of various RT methods and provides insights into how RT modulates innate immune signaling.
Keywords: DNA damage; NK cells; dendric cells; hypoxia; macrophages; microbubble oxygen; radiation therapy; tumor microenvironment.
Conflict of interest statement
The authors have no conflict of interest.
Figures


Similar articles
-
Targeting Innate Immunity to Enhance the Efficacy of Radiation Therapy.Front Immunol. 2019 Jan 14;9:3077. doi: 10.3389/fimmu.2018.03077. eCollection 2018. Front Immunol. 2019. PMID: 30692991 Free PMC article. Review.
-
Immunomodulatory Effects of Radiotherapy.Int J Mol Sci. 2020 Oct 31;21(21):8151. doi: 10.3390/ijms21218151. Int J Mol Sci. 2020. PMID: 33142765 Free PMC article. Review.
-
Radiation therapy and the innate immune response: Clinical implications for immunotherapy approaches.Br J Clin Pharmacol. 2020 Sep;86(9):1726-1735. doi: 10.1111/bcp.14351. Epub 2020 Jun 3. Br J Clin Pharmacol. 2020. PMID: 32388875 Free PMC article. Review.
-
Modulating Both Tumor Cell Death and Innate Immunity Is Essential for Improving Radiation Therapy Effectiveness.Front Immunol. 2017 May 26;8:613. doi: 10.3389/fimmu.2017.00613. eCollection 2017. Front Immunol. 2017. PMID: 28603525 Free PMC article. Review.
-
Targeting Innate Immunity in Cancer Therapy.Vaccines (Basel). 2021 Feb 9;9(2):138. doi: 10.3390/vaccines9020138. Vaccines (Basel). 2021. PMID: 33572196 Free PMC article. Review.
Cited by
-
Results of an Integrated Phase I/II Prospective Clinical Trial (NEXIS) for Neoadjuvant Anti-PD-L1 (Durvalumab) and Anti-CTLA-4 (Tremelimumab) With Radiation for High-Risk Soft-Tissue Sarcoma of the Trunk and Extremities.Cureus. 2024 Oct 22;16(10):e72119. doi: 10.7759/cureus.72119. eCollection 2024 Oct. Cureus. 2024. PMID: 39575028 Free PMC article.
-
Therapeutic In Situ Cancer Vaccine Using Pulsed Stereotactic Body Radiotherapy-A Translational Model.Vaccines (Basel). 2023 Dec 20;12(1):7. doi: 10.3390/vaccines12010007. Vaccines (Basel). 2023. PMID: 38276666 Free PMC article.
-
Consensus, debate, and prospective on pancreatic cancer treatments.J Hematol Oncol. 2024 Oct 10;17(1):92. doi: 10.1186/s13045-024-01613-x. J Hematol Oncol. 2024. PMID: 39390609 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

