Matching-Adjusted Indirect Comparison of the Long-Term Efficacy of Deucravacitinib Versus Adalimumab for Moderate to Severe Plaque Psoriasis
- PMID: 37525000
- PMCID: PMC10613163
- DOI: 10.1007/s13555-023-00977-1
Matching-Adjusted Indirect Comparison of the Long-Term Efficacy of Deucravacitinib Versus Adalimumab for Moderate to Severe Plaque Psoriasis
Erratum in
-
Correction to: Matching-Adjusted Indirect Comparison of the Long-Term Efficacy of Deucravacitinib Versus Adalimumab for Moderate to Severe Plaque Psoriasis.Dermatol Ther (Heidelb). 2023 Nov;13(11):2605-2607. doi: 10.1007/s13555-023-01043-6. Dermatol Ther (Heidelb). 2023. PMID: 37853229 Free PMC article. No abstract available.
Abstract
Introduction: Deucravacitinib, an oral tyrosine kinase 2 (TYK2) inhibitor, is approved in the United States to treat adults with moderate-to-severe plaque psoriasis (PsO). This study compared the long-term efficacy of deucravacitinib and adalimumab using results from long-term extension (LTE) trials.
Methods: Open-label LTE trials were identified for an indirect treatment comparison (deucravacitinib: POETYK PSO-LTE [NCT04036435]; adalimumab: REVEAL extension [NCT00195676]). To ensure study design comparability, patients initially randomized to placebo and switched to deucravacitinib or adalimumab after week 16 were compared. The primary outcome was an ≥ 75% reduction in Psoriasis Area and Severity Index score (PASI 75) at week 112 postrandomization. Secondary outcomes were PASI 75 at week 52 and an ≥ 90% reduction in PASI score (PASI 90) at weeks 52 and 112. Missing PASI data were imputed. A matching-adjusted indirect comparison was conducted; individual patient-level data from POETYK PSO-LTE were reweighted to balance baseline characteristics with those from the REVEAL extension.
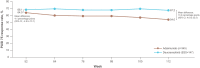
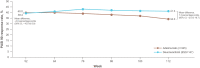
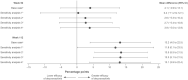
Results: Before reweighting, on average, patients in the POETYK PSO-LTE (N = 329) versus the REVEAL (N = 345) extension were older, had a lower body weight, received more prior systemic treatments, and had higher baseline PASI scores and week 16 placebo PASI 75 and PASI 90 response rates. Following reweighting, adjusted week 112 PASI 75 response rates were significantly higher for deucravacitinib versus adalimumab (67.2% vs. 54.0%; mean difference [95% CI], 13.2 [4.0-22.5] percentage points). Deucravacitinib had a numerically higher adjusted week 112 PASI 90 response rate (41.3% vs. 34.0%; mean difference [95% CI], 7.3 [-2.0 to 16.7] percentage points). The treatments had similar week 52 adjusted PASI 75 and PASI 90 response rates.
Conclusion: In this interim analysis, adults with moderate to severe PsO had higher long-term response rates at 2 years when treated with deucravacitinib versus adalimumab. Deucravacitinib response rates remained stable whereas adalimumab response rates declined in year 2.
Keywords: Adalimumab; Deucravacitinib; Indirect comparison; MAIC; Plaque psoriasis.
Plain language summary
Plaque psoriasis is an inflammatory disease that causes red, itchy, dry patches (called plaques) on the skin. The disease cannot be cured, but the symptoms can be treated. Deucravacitinib and adalimumab are two treatments approved for use in adults with moderate to severe plaque psoriasis; deucravacitinib is an oral medication and adalimumab is injected with a needle under the skin. Each treatment has proven its efficacy compared with placebo (a pill or injection with no active effect) in separate clinical trials, but because no two clinical trials are exactly alike, the results cannot be accurately compared. Matching-adjusted indirect comparison is a method used to compare the results of one clinical trial with those of another when a direct comparison is not possible; characteristics from the patients in one trial are made to match the patient population in the other trial, and the adjusted results are compared. We performed a matching-adjusted indirect comparison of an open-label extension trial of deucravacitinib with an open-label extension trial of adalimumab to study the long-term efficacy of each treatment. At 1 year of treatment, we observed that similar proportions of patients receiving each treatment achieved a 75% or 90% improvement from their baseline Psoriasis Area and Severity Index score, called PASI 75 or PASI 90, respectively. At 2 years of treatment, similar proportions achieved PASI 90, but the proportion of patients receiving deucravacitinib who achieved PASI 75 was greater than that of patients receiving adalimumab.
© 2023. The Author(s).
Conflict of interest statement
April W. Armstrong has received research grants and personal fees from Bristol Myers Squibb, Eli Lilly, Janssen, Leo Pharma, and Novartis; personal fees from Boehringer Ingelheim/Parexel, Celgene, Dermavant, Genentech, GlaxoSmithKline, Menlo Therapeutics, Merck, Modernizing Medicine, Ortho Dermatologics, Pfizer, Regeneron, Sanofi Genzyme, Science 37, Sun Pharma, and Valeant; and grants from Dermira, Kyowa Hakko Kirin, and UCB outside the submitted work. Sang Hee Park and Vardhaman Patel are employees of and may own stock options in Bristol Myers Squibb. David Davidson was an employee of Bristol Myers Squibb at the time of this study. Malcolm Hogan is an employee of Syneos Health, which provides services to Bristol Myers Squibb. Viktor Chirikov and Wei-Jhih Wang are employees of OPEN Health Evidence & Access, which was contracted by Bristol Myers Squibb to support this research.
Figures

Similar articles
-
Deucravacitinib in plaque psoriasis: 2-year safety and efficacy results from the phase III POETYK trials.Br J Dermatol. 2024 Apr 17;190(5):668-679. doi: 10.1093/bjd/ljae014. Br J Dermatol. 2024. PMID: 38226713 Clinical Trial.
-
Safety and Efficacy of Deucravacitinib in Moderate to Severe Plaque Psoriasis for Up to 3 Years: An Open-Label Extension of Randomized Clinical Trials.JAMA Dermatol. 2025 Jan 1;161(1):56-66. doi: 10.1001/jamadermatol.2024.4688. JAMA Dermatol. 2025. PMID: 39602111 Free PMC article. Clinical Trial.
-
Efficacy and safety of the selective TYK2 inhibitor, deucravacitinib, in Japanese patients with moderate to severe plaque psoriasis: Subgroup analysis of a randomized, double-blind, placebo-controlled, global phase 3 trial.J Dermatol. 2023 May;50(5):588-595. doi: 10.1111/1346-8138.16740. Epub 2023 Mar 7. J Dermatol. 2023. PMID: 36882942 Clinical Trial.
-
Deucravacitinib: The First FDA-Approved Oral TYK2 Inhibitor for Moderate to Severe Plaque Psoriasis.Ann Pharmacother. 2024 Apr;58(4):416-427. doi: 10.1177/10600280231153863. Epub 2023 Jun 21. Ann Pharmacother. 2024. PMID: 37341177 Review.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2022 May 23;5(5):CD011535. doi: 10.1002/14651858.CD011535.pub5. Cochrane Database Syst Rev. 2022. Update in: Cochrane Database Syst Rev. 2023 Jul 12;7:CD011535. doi: 10.1002/14651858.CD011535.pub6. PMID: 35603936 Free PMC article. Updated. Review.
Cited by
-
Pharmacological inhibition of tyrosine protein-kinase 2 reduces islet inflammation and delays type 1 diabetes onset in mice.bioRxiv [Preprint]. 2024 May 9:2024.03.20.585925. doi: 10.1101/2024.03.20.585925. bioRxiv. 2024. PMID: 38766166 Free PMC article. Preprint.
-
New and Emerging Oral/Topical Small-Molecule Treatments for Psoriasis.Pharmaceutics. 2024 Feb 6;16(2):239. doi: 10.3390/pharmaceutics16020239. Pharmaceutics. 2024. PMID: 38399292 Free PMC article. Review.
-
New and emerging oral therapies for psoriasis.Drugs Context. 2024 Aug 1;13:2024-5-6. doi: 10.7573/dic.2024-5-6. eCollection 2024. Drugs Context. 2024. PMID: 39131603 Free PMC article. Review.
-
Matching-Adjusted Indirect Comparison of Risankizumab Versus Deucravacitinib in Patients with Moderate-to-Severe Plaque Psoriasis.Dermatol Ther (Heidelb). 2024 Nov;14(11):3071-3081. doi: 10.1007/s13555-024-01293-y. Epub 2024 Oct 25. Dermatol Ther (Heidelb). 2024. PMID: 39453596 Free PMC article.
References
-
- National Psoriasis Foundation. About psoriasis. 2022. https://www.psoriasis.org/about-psoriasis/. Accessed 7 Feb 2023.
-
- Wu JJ. Contemporary management of moderate to severe plaque psoriasis. Am J Manag Care. 2017;23(21 Suppl):S403–S416. - PubMed
-
- Humira (package insert). North Chicago, IL: Abbott Laboratories (AbbVie); 2022.
LinkOut - more resources
Full Text Sources

