Effective GH Replacement With Somapacitan in Children With GHD: REAL4 2-year Results and After Switch From Daily GH
- PMID: 37406251
- PMCID: PMC10655534
- DOI: 10.1210/clinem/dgad394
Effective GH Replacement With Somapacitan in Children With GHD: REAL4 2-year Results and After Switch From Daily GH
Erratum in
-
Correction to: "Effective GH Replacement With Somapacitan in Children With GHD: REAL4 2-year Results and After Switch From Daily GH".J Clin Endocrinol Metab. 2023 Nov 17;108(12):e1762. doi: 10.1210/clinem/dgad497. J Clin Endocrinol Metab. 2023. PMID: 37619571 Free PMC article. No abstract available.
Abstract
Context: Somapacitan is a long-acting GH derivative for treatment of GH deficiency (GHD).
Objective: Evaluate the efficacy and tolerability of somapacitan in children with GHD after 2 years of treatment and after the switch from daily GH.
Design: A randomized, multinational, open-labelled, controlled parallel group phase 3 trial, comprising a 52-week main phase and 3-year safety extension (NCT03811535).
Setting: Eighty-five sites across 20 countries.
Patients: A total of 200 treatment-naïve prepubertal patients were randomized and exposed; 194 completed the 2-year period.
Interventions: Patients were randomized 2:1 to somapacitan (0.16 mg/kg/wk) or daily GH (0.034 mg/kg/d) during the first year, after which all patients received somapacitan 0.16 mg/kg/wk.
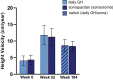
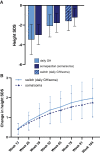
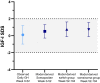
Main outcome measures: Height velocity (HV; cm/year) at week 104. Additional assessments included HV SD score (SDS), height SDS, IGF-I SDS, and observer-reported outcomes.
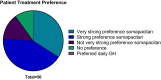
Results: HV was sustained in both groups between 52 and 104 weeks. At week 104, mean (SD) for HV between weeks 52 and 104 was 8.4 (1.5) cm/year after continuous somapacitan treatment and 8.7 (1.8) cm/year after 1 year of somapacitan treatment following switch from daily GH. Secondary height-related endpoints also supported sustained growth. Mean IGF-I SDS during year 2 was similar between groups and within normal range (-2 to +2). Somapacitan was well tolerated, with no safety or tolerability issues identified. GH patient preference questionnaire results show that most patients and their caregivers (90%) who switched treatment at year 2 preferred once-weekly somapacitan over daily GH treatment.
Conclusions: Somapacitan in children with GHD showed sustained efficacy and tolerability for 2 years, and after switching from daily GH. Patients/caregivers switching from daily GH expressed a preference for somapacitan.
Clinical trial registration: NCT03811535.
Keywords: growth hormone; growth hormone deficiency; growth hormone replacement therapy; long-acting growth hormone; somapacitan.
© The Author(s) 2023. Published by Oxford University Press on behalf of the Endocrine Society.
Figures
Similar articles
-
Falls prevention interventions for community-dwelling older adults: systematic review and meta-analysis of benefits, harms, and patient values and preferences.Syst Rev. 2024 Nov 26;13(1):289. doi: 10.1186/s13643-024-02681-3. Syst Rev. 2024. PMID: 39593159 Free PMC article.
-
Effective growth hormone replacement with once-weekly somapacitan in Japanese children with growth hormone deficiency: Results from REAL4, a phase 3 clinical trial.Clin Endocrinol (Oxf). 2024 Apr;100(4):389-398. doi: 10.1111/cen.15025. Epub 2024 Feb 18. Clin Endocrinol (Oxf). 2024. PMID: 38368603 Clinical Trial.
-
Safety, pharmacokinetics, and early bactericidal activity of quabodepistat in combination with delamanid, bedaquiline, or both in adults with pulmonary tuberculosis: a randomised, active-controlled, open-label trial.Lancet Infect Dis. 2024 Nov 26:S1473-3099(24)00601-7. doi: 10.1016/S1473-3099(24)00601-7. Online ahead of print. Lancet Infect Dis. 2024. PMID: 39612928
-
Interventions for supporting pregnant women's decision-making about mode of birth after a caesarean.Cochrane Database Syst Rev. 2013 Jul 30;2013(7):CD010041. doi: 10.1002/14651858.CD010041.pub2. Cochrane Database Syst Rev. 2013. PMID: 23897547 Free PMC article. Review.
-
Methods of milk expression for lactating women.Cochrane Database Syst Rev. 2016 Sep 29;9(9):CD006170. doi: 10.1002/14651858.CD006170.pub5. Cochrane Database Syst Rev. 2016. PMID: 27684560 Free PMC article. Review.
Cited by
-
Diagnosing and treating anterior pituitary hormone deficiency in pediatric patients.Rev Endocr Metab Disord. 2024 Jun;25(3):555-573. doi: 10.1007/s11154-023-09868-4. Epub 2023 Dec 19. Rev Endocr Metab Disord. 2024. PMID: 38112850 Review.
-
Once-Weekly Somapacitan as an Alternative Management of Growth Hormone Deficiency in Prepubertal Children: A Systematic Review and Meta-Analysis of Randomized Controlled Trial.Children (Basel). 2024 Feb 9;11(2):227. doi: 10.3390/children11020227. Children (Basel). 2024. PMID: 38397339 Free PMC article. Review.
-
Developments in the Management of Growth Hormone Deficiency: Clinical Utility of Somapacitan.Drug Des Devel Ther. 2024 Feb 3;18:291-306. doi: 10.2147/DDDT.S315172. eCollection 2024. Drug Des Devel Ther. 2024. PMID: 38333899 Free PMC article. Review.
-
Investigating the Bioavailability and Insulin-like Growth Factor-I Release of Two Different Strengths of Somapacitan: A Randomised, Double-Blind Crossover Trial.Clin Pharmacokinet. 2024 Jul;63(7):1015-1024. doi: 10.1007/s40262-024-01395-y. Epub 2024 Jul 5. Clin Pharmacokinet. 2024. PMID: 38969919 Free PMC article. Clinical Trial.
-
Efficacy and Safety of Somapacitan Relative to Somatrogon and Lonapegsomatropin in Pediatric Growth Hormone Deficiency: Systematic Literature Review and Network Meta-analysis.Adv Ther. 2024 Nov;41(11):4098-4124. doi: 10.1007/s12325-024-02966-y. Epub 2024 Sep 11. Adv Ther. 2024. PMID: 39261416 Free PMC article.
References
-
- Yuen KCJ, Miller BS, Biller BMK. The current state of long-acting growth hormone preparations for growth hormone therapy. Curr Opin Endocrinol Diabetes Obes. 2018;25(4):267‐273. - PubMed

