Seladelpar efficacy and safety at 3 months in patients with primary biliary cholangitis: ENHANCE, a phase 3, randomized, placebo-controlled study
- PMID: 37386786
- PMCID: PMC10344437
- DOI: 10.1097/HEP.0000000000000395
Seladelpar efficacy and safety at 3 months in patients with primary biliary cholangitis: ENHANCE, a phase 3, randomized, placebo-controlled study
Abstract
Background and aims: ENHANCE was a phase 3 study that evaluated efficacy and safety of seladelpar, a selective peroxisome proliferator-activated receptor-δ (PPAR) agonist, versus placebo in patients with primary biliary cholangitis with inadequate response or intolerance to ursodeoxycholic acid (UDCA).
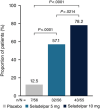
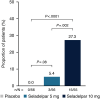
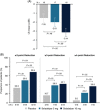
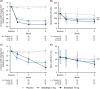
Approach and results: Patients were randomized 1:1:1 to oral seladelpar 5 mg (n=89), 10 mg (n=89), placebo (n=87) daily (with UDCA, as appropriate). Primary end point was a composite biochemical response [alkaline phosphatase (ALP) < 1.67×upper limit of normal (ULN), ≥15% ALP decrease from baseline, and total bilirubin ≤ ULN] at month 12. Key secondary end points were ALP normalization at month 12 and change in pruritus numerical rating scale (NRS) at month 6 in patients with baseline score ≥4. Aminotransferases were assessed. ENHANCE was terminated early following an erroneous safety signal in a concurrent, NASH trial. While blinded, primary and secondary efficacy end points were amended to month 3. Significantly more patients receiving seladelpar met the primary end point (seladelpar 5 mg: 57.1%, 10 mg: 78.2%) versus placebo (12.5%) ( p < 0.0001). ALP normalization occurred in 5.4% ( p =0.08) and 27.3% ( p < 0.0001) of patients receiving 5 and 10 mg seladelpar, respectively, versus 0% receiving placebo. Seladelpar 10 mg significantly reduced mean pruritus NRS versus placebo [10 mg: -3.14 ( p =0.02); placebo: -1.55]. Alanine aminotransferase decreased significantly with seladelpar versus placebo [5 mg: 23.4% ( p =0.0008); 10 mg: 16.7% ( p =0.03); placebo: 4%]. There were no serious treatment-related adverse events.
Conclusions: Patients with primary biliary cholangitis (PBC) with inadequate response or intolerance to UDCA who were treated with seladelpar 10 mg had significant improvements in liver biochemistry and pruritus. Seladelpar appeared safe and well tolerated.
Copyright © 2023 The Author(s). Published by Wolters Kluwer Health, Inc.
Conflict of interest statement
Gideon M. Hirschfield consults for and received payment/honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from GlaxoSmithKline, Intercept Pharma, and Ipsen. He consults for CymaBay Therapeutics, Escient, Gilead, Mirum, and Pliant. Mitchell L. Shiffman consults for, is on the speakers’ bureau for, and received grants from Intercept. He consults for and is on the speakers’ bureau for Intra-Sana. He is on the speakers’ bureau for and received grants from Cymbay. He received grants from Genfit, Hightide, and Mirum. Aliya Gulamhusein consults for and is on the speakers’ bureau for Intercept. She consults for CymaBay. Kris V. Kowdley consults for, is on the speakers’ bureau for and received grants from 89 Bio, Genfit, Gilead, Intercept, and CymbaBay. He consults for and received grants from Madrigal, Mirum, NGM, Pliant, and Zydus. He consults for and owns stock in Inipharm. He consults for Calliditas and Ipsen. He is on the speakers’ bureau for AbbVie. He received grants from GSK, Pfizer, Hanmi, HighTide, Viking, and Janssen. He receives royalties from UpToDate. He received payment for expert testimony from the Department of Justice. He participated in a data safety monitoring board or advisory board for CTI, Durect, and Labcorp. He received equipment, materials, drugs, medical writing, gifts, or other services from Sonic Insight. John M. Vierling advises and received grants from CymaBay, Intercept, Lilly, Novartis, and Sagiment. He advises Arena, Blade, Kezar, Labcorp, Fractyl, Ipsen, Moderna, and Taiwan J. He received grants from Genfit. He is a board member of and owns stock in Athenex. Cynthia Levy consults for, advises, and received grants from Cara Therapeutics, CymaBay Therapeutics, and GlaxoSmith Kline. She consults for and received grants from Calliditas, Escient, Genfit, Gilead, Intercept, Ipsen, Mirum, and Target RWE. She received grants from HighTide, Novartis, and Zydus. Andreas E. Kremer consults for, received payment/honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from, received grants from, and advises AbbVie, Bayer, CymaBay Therapeutics, Gilead, GlaxoSmithKline, Intercept Pharma, and MSD. He received payment/honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from and received grants from AOP Orphan, Bristol Myers Squibb, CMS, Dr. Falk, Eisai, Eli Lily, Janssen, Newbridge, Novartis, and Zambon. He consults for and advises Beiersdorf, Escient, FMC, Guidepoint, Medscape, Mirum, Myr, Roche, and Viofor. Ehud Zigmond advises, received lecture fees from, and received grants from Neopharm LTD. Stuart C. Gordon consults for and received grants from CymaBay, Gilead, and GSK. He received grants from AbbVie, DURECT, Genfit, Hightide, Intercept Pharma, Merck, Mirum, Pliant, and Viking. Christopher L. Bowlus consults for and received grants from CymaBay Therapeutics, Eli Lilly, and GlaxoSmithKline. He consults for BiomX, Mirum, Shire, and Trevi Therapeutics. He received grants from Arena Pharmaceuticals, Bristol Myers Squibb, Calliditas Therapeutics, Chemomab, COUR Pharmaceuticals, Genfit, Gilead, GlaxoSmithKline, Hanmi, Intercept, Novartis, NovoNordisk, Pliant, Takeda, and TARGET. Eric J. Lawitz consults for, is on the speakers’ bureau for, and received grants from Intercept. He consults for and received grants from Akero, Boheringer Ingelheim, BMS, Novo Nordisk, Metacrine, Sagimet, and Terns. He is on the speakers’ bureau for and received grants from AbbVie and Gilead. He received grants from 89Bio Inc., Allergan, Alnylam Pharmaceuticals Inc., Amgen, Ascelia Pharma, Assemblybio, AstraZeneca, Axcella Health, Biocryst Pharmaceuticals, Bird Rock Bio Inc., Conatus Pharmaceuticals, CymaBay Therapeutics, CytoDyn, DSM, Durect Corporation, Eli Lilly, Enanta Pharmaceuticals, Enyo Pharma, Exalenz Bioscience, Galectin Therapeutics, Galmed Pharmaceuticals, Genentech, Genfit, GlaxoSmithKline, Hanmi Pharmaceuticals, HighTide Biopharma, Inventiva, Janssen Pharmaceuticals, Laboratory for Advanced Medicine, Loxo Oncology, Madrigal Pharmaceuticals, Merck, NGM Biopharmaceuticals Inc., Northsea Therapeutics, Novartis, Pfizer, Poxel Co., Roche, Synlogic Therapeutics, Viking Therapeutics, and Zydus Pharmaceuticals. Richard J. Aspinall consults for and is on the speakers’ bureau for Norgine UK. He is on the speakers’ bureau for Intercept UK. He owns stock in CymaBay. Daniel S. Pratt consults for Mediar Therapeutics. Karina Raikhelson received payment/honoraria for lectures presentations, speakers bureaus, manuscript writing, or educational events from Abbott Laboratories GmbH, Alfasigma Rus, Binnopharm Group, Dr. Falk Pharma GmbH, JSC Nizhfarm, and JSC Pharmstandard. She received payment/honoraria for lectures presentations, speakers bureaus, manuscript writing, or educational events and was expert witness for STPF “Polisan.” She received grants from CymaBay and Novartis. Michael A. Heneghan consulting for Ipsen, Eledon, and Moderna He recieved payment/honoraria for lectures, presentations, speakers bureau, manuscript writing, or educational events from Falk, Advanz Pharma, and Intercept Pharma. He receives royalties from UpToDate. Sook-Hyang Jeong received grants from Bristol Myers Squibb, CymaBay Therapeutics, Galmed, Gilead, Hoffmann-La Roche Ltd, Intercept, and MSD. Alma L. Ladrón de Guevara received grants from AstraZeneca, Cymabay, Lilly, Madrigal, Akero, Galectin, MSD and Novo Nordisk, and Viking. Marlyn J. Mayo consults for and received grants from CymaBay Therapeutics, GlaxoSmithKline, and Mallinckrodt. She received grants from Genfit, Intercept, Mirum, and TARGET. She consults for Ipsen. She is on the speaker’s bureau for Intra-Sana. George N. Dalekos advises, is on the speakers’ bureau for, and serves as principal investigator for Genkeyotex, Pfizer, and Sobi. He advises and is on the speakers’ bureau for Ipsen and Sanofi. He is a principal investigator for Amyndas Pharmaceuticals, CymBay Therapeutics, Intercept, Novo Nordisk, Regulus Therapeutics, and Tiziana Life Sciences. Joost P.H. Drenth consults for Camurus. He received grants from AbbVie and Gilead. He is on a data safety monitoring board or advisory board for COIN B Study. Ewa Janczewska advises and received grants from Novo Nordisk and Cellaion. She is on the speakers’ bureau for and received grants from Abbvie. She is on the speakers’ bureau for Roche. She received grants from Axella, Exelixis, CymaBay, Calliditas, BMS, GSK, Janssen-Cilag, Dr. Falk, Inventiva, and MSD. Barbara A. Leggett received grants from CymaBay Therapeutics. Frederik Nevens consults for and received grants from Gilead. He consults for Abbvie, W.L. Gore, Cook Medical, TwinPharma, Intercept, Genkyotex, Camurus, Chemomab Therapeutics, Agomab Therapeutics, Novartis Pharma, Mayoly Spindler, Calliditas Therapeutics, Norgine, Takeda, and Dynacure. He received grants from Promethera Therapeutics and Ipsen. Victor Vargas consults for Genfit. He is on the speakers’ bureau for Intercept. He is on the data safety monitoring board or advisory board for Promethera Biosciences/Cellaion. He received grants from Advanz Pharma. Christophe Corpechot consults for and received grants from Intercept and CymaBay. He consults for Genkyotex/Calliditas. He received grants from Arrow Génériques, Biotest, and Gilead. Eduardo L. Fassio received grants from Gador. Eli Zuckerman consults and is a speaker for AbbVie, Gilead, GlaxoSmithKline, Janssen, Merck, Neopharm, NovoNordisk, and Roche. He is also a speaker for Bristol Meyers Squibb and Novartis. Holger Hinrichsen consults for and received payment/honoraria for lectures, presentations, and speakers bureaus from AbbVie, Gilead, and Intercept Pharma. He received payment/honoraria for lectures, presentations, and speakers bureaus Norgine. Pietro Invernizzi advises, is on the speakers’ bureau for, and received grants from Intercept. He advises Calliditas and Zydus. He received grants from Abbvie. Palak J. Trivedi consults for and advises CymaBay. David E.J. Jones consults for, is on the speakers’ bureau for, and received funding from Intercept. He consults for CymaBay Therapeutics, Kowa, and Umecrine. He is on the speakers’ bureau for Falk, GlaxoSmithKline, and Ipsen. Mark G. Swain consults for, is on the speakers’ bureau for, and received grants from Gilead. He consults for and received grants from Pfizer and Novartis. He consults for Ipsen. He is on the speakers’ bureau for Abbott. He received grants from AbbVie, Ancella, AstraZeneca, Bristol Myers Squibb, Calliditas Therapeutics, Celgene, CymaBay Therapeutics, Galectin, Genfit, GlaxoSmithKline, Intercept, and Novo Nordisk. Alexandra Steinberg owns stock, has intellectual property rights in and was employed by CymaBay Therapeutics when the work was conducted, and is employed by Carmot Therapeutics. Pol F. Boudes was employed by CymaBay Therapeutics when the work was conducted and declares a seladelpar method of use patent for PBC. He is currently employed by Galectin Therapeutics. Yun-Jung Choi is employed by CymaBay Therapeutics. Charles A. McWherter owns stock and intellectual property rights in and is employed by CymaBay. He declares a seladelpar method of use patent for PBC and for cholestatic pruritus. The remaining authors have no conflicts to report.
Figures

Comment in
-
PPARα, PPARδ, or both-that is the question!Hepatology. 2024 Jul 1;80(1):8-10. doi: 10.1097/HEP.0000000000000791. Epub 2024 Feb 16. Hepatology. 2024. PMID: 38373084 No abstract available.
Similar articles
-
A Phase 3 Trial of Seladelpar in Primary Biliary Cholangitis.N Engl J Med. 2024 Feb 29;390(9):783-794. doi: 10.1056/NEJMoa2312100. Epub 2024 Feb 21. N Engl J Med. 2024. PMID: 38381664 Clinical Trial.
-
A phase II, randomized, open-label, 52-week study of seladelpar in patients with primary biliary cholangitis.J Hepatol. 2022 Aug;77(2):353-364. doi: 10.1016/j.jhep.2022.02.033. Epub 2022 Mar 30. J Hepatol. 2022. PMID: 35367282 Clinical Trial.
-
Seladelpar (MBX-8025), a selective PPAR-δ agonist, in patients with primary biliary cholangitis with an inadequate response to ursodeoxycholic acid: a double-blind, randomised, placebo-controlled, phase 2, proof-of-concept study.Lancet Gastroenterol Hepatol. 2017 Oct;2(10):716-726. doi: 10.1016/S2468-1253(17)30246-7. Epub 2017 Aug 14. Lancet Gastroenterol Hepatol. 2017. PMID: 28818518 Clinical Trial.
-
Evaluating the safety and efficacy of seladelpar for adults with primary biliary cholangitis.Expert Opin Pharmacother. 2024 Aug;25(11):1517-1523. doi: 10.1080/14656566.2024.2390120. Epub 2024 Aug 11. Expert Opin Pharmacother. 2024. PMID: 39107982 Review.
-
Obeticholic acid for the treatment of primary biliary cholangitis.Expert Opin Pharmacother. 2016 Sep;17(13):1809-15. doi: 10.1080/14656566.2016.1218471. Epub 2016 Aug 9. Expert Opin Pharmacother. 2016. PMID: 27468093 Review.
Cited by
-
Managing pruritus in chronic liver disease: An in-depth narrative review.Clin Liver Dis (Hoboken). 2024 Jun 12;23(1):e0187. doi: 10.1097/CLD.0000000000000187. eCollection 2024 Jan-Jun. Clin Liver Dis (Hoboken). 2024. PMID: 38872783 Free PMC article. Review. No abstract available.
-
Itching for Answers: A Comprehensive Review of Cholestatic Pruritus Treatments.Biomolecules. 2024 Sep 28;14(10):1227. doi: 10.3390/biom14101227. Biomolecules. 2024. PMID: 39456160 Free PMC article. Review.
-
Seladelpar: First Approval.Drugs. 2024 Nov;84(11):1487-1495. doi: 10.1007/s40265-024-02114-4. Epub 2024 Nov 22. Drugs. 2024. PMID: 39572508 Review.
-
AI-based automation of enrollment criteria and endpoint assessment in clinical trials in liver diseases.Nat Med. 2024 Oct;30(10):2914-2923. doi: 10.1038/s41591-024-03172-7. Epub 2024 Aug 7. Nat Med. 2024. PMID: 39112795 Free PMC article.
-
Seladelpar treatment reduces IL-31 and pruritus in patients with primary biliary cholangitis.Hepatology. 2024 Jul 1;80(1):27-37. doi: 10.1097/HEP.0000000000000728. Epub 2023 Dec 20. Hepatology. 2024. PMID: 38117036 Free PMC article. Clinical Trial.
References
-
- Trivedi PJ, Hirschfield GM. Recent advances in clinical practice: epidemiology of autoimmune liver diseases. Gut. 2021;70:1989–2003. - PubMed
-
- Lleo A, Wang GQ, Gershwin ME, Hirschfield GM. Primary biliary cholangitis. Lancet. 2020;396:1915–26. - PubMed
-
- Gulamhusein AF, Hansen BE. Beyond biochemical responses, use of histologic staging to predict outcomes of patients with primary biliary cholangitis. Clin Gastroenterol Hepatol. 2020;18:1033–5. - PubMed
-
- Lindor KD, Bowlus CL, Boyer J, Levy C, Mayo M. Primary biliary cholangitis: 2018 practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2019;69:394–419. - PubMed

