What are the benefits of the anti-FGF23 antibody burosumab on the manifestations of X-linked hypophosphatemia in adults in comparison with conventional therapy? A review
- PMID: 37180412
- PMCID: PMC10032432
- DOI: 10.1177/26330040221074702
What are the benefits of the anti-FGF23 antibody burosumab on the manifestations of X-linked hypophosphatemia in adults in comparison with conventional therapy? A review
Abstract
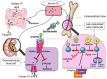
X-linked hypophosphatemia (XLH) is a genetic disease mostly related to PHEX gene mutations which increases FGF23 serum levels, leading to hypophosphatemia and osteomalacia in adults, while affected children, in addition, develop rickets. Most of adults with XLH suffer from reduced quality of life and physical disability due to chronic bone and joint pain related to limb deformities, early osteoarthritis, delayed-healing of insufficiency fractures, and enthesopathies. Dental infections, muscle dysfunction, and deafness are also frequent. The current treatment consists of 2-5 times daily oral administration of phosphate combined to active vitamin D, often badly tolerated with immediate digestive side effects, responsible for poor compliance. In the long term, it may induce nephrocalcinosis and hyperparathyroidism. Burosumab, an anti-FGF23 blocking antibody, was approved for treating children with XLH in many countries. A randomized 24-week-long placebo-controlled trial, followed by an open-label period of equal duration was conducted in 134 XLH adults treated with 1 mg/kg burosumab/4 weeks. During burosumab treatment, 94% of the patients normalized serum phosphate values versus 7% in the placebo group. Fracture healing was increased 16.7 times compared with placebo-treated patients. All pain and disability tests improved significantly in a time-dependent manner. Burosumab for 48 weeks improved histological lesions of osteomalacia in a single-arm longitudinal study analyzing paired bone biopsies. Another single-arm, open-label study investigated the long-term safety and efficacy of burosumab in 20 adult patients followed for 3.2 years. Burosumab was beneficial on pain and disability scores and on bone remodeling markers. No major side effects especially no hyperphosphatemic episodes were reported. Overall, the benefit/risk ratio of burosumab is positive in adult patients with clinical and/or biological complications of XLH. Burosumab corrects hypophosphatemia, promotes fracture healing, and induces a modest but significant effect on XLH-induced subjective pain and disability symptoms.
Plain language title and summary: Effects of conventional treatment and burosumab in adults with X-linked hypophosphatemia.X-linked hypophosphatemia (XLH) is a disease of genetic origin that affects mineralized tissues (skeleton and teeth) and impairs muscle function. It induces a decrease in blood phosphate levels. This leads to under mineralization of bones and insufficiency fractures that heal slowly, associated with poor dental health characterized by spontaneous dental abscesses. Adults with XLH suffer from chronic pain and limb deformities that alter their quality of life. They are currently treated with daily administration of vitamin D and several daily doses of phosphate. This treatment may induce parathyroid gland dysfunction and mineral deposits in the kidney. If not tightly monitored, these side effects may lead to tertiary hyperparathyroidism and the need for parathyroid gland surgery, or to nephrocalcinosis which may proceed to chronic kidney disease. Burosumab is an antibody that blocks the action of FGF23 the factor that circulates in excess in blood and is responsible for phosphate renal leak in XLH. Three studies demonstrated that burosumab, injected every 4 weeks, is efficient and safe for treating adults with XLH.
Keywords: FGF23; adults; burosumab; hypophosphatemia.
© The Author(s), 2022.
Conflict of interest statement
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures
Similar articles
-
A Randomized, Double-Blind, Placebo-Controlled, Phase 3 Trial Evaluating the Efficacy of Burosumab, an Anti-FGF23 Antibody, in Adults With X-Linked Hypophosphatemia: Week 24 Primary Analysis.J Bone Miner Res. 2018 Aug;33(8):1383-1393. doi: 10.1002/jbmr.3475. Epub 2018 Jun 26. J Bone Miner Res. 2018. PMID: 29947083 Clinical Trial.
-
Safety and efficacy of burosumab in improving phosphate metabolism, bone health, and quality of life in adolescents with X-linked hypophosphatemic rickets.Eur J Med Genet. 2024 Aug;70:104958. doi: 10.1016/j.ejmg.2024.104958. Epub 2024 Jun 29. Eur J Med Genet. 2024. PMID: 38950880
-
Bone Matrix Mineralization and Response to Burosumab in Adult Patients With X-Linked Hypophosphatemia: Results From the Phase 3, Single-Arm International Trial.J Bone Miner Res. 2022 Sep;37(9):1665-1678. doi: 10.1002/jbmr.4641. Epub 2022 Aug 10. J Bone Miner Res. 2022. PMID: 35775373 Clinical Trial.
-
Burosumab for Pediatric X-Linked Hypophosphatemia.Curr Osteoporos Rep. 2021 Jun;19(3):271-277. doi: 10.1007/s11914-021-00669-9. Epub 2021 May 10. Curr Osteoporos Rep. 2021. PMID: 33970403 Free PMC article. Review.
-
Phosphatonins: From Discovery to Therapeutics.Endocr Pract. 2023 Jan;29(1):69-79. doi: 10.1016/j.eprac.2022.09.007. Epub 2022 Oct 7. Endocr Pract. 2023. PMID: 36210014 Review.
Cited by
-
Dental impact of anti-fibroblast growth factor 23 therapy in X-linked hypophosphatemia.Int J Oral Sci. 2023 Dec 6;15(1):53. doi: 10.1038/s41368-023-00259-8. Int J Oral Sci. 2023. PMID: 38052774 Free PMC article.
-
Fibroblast Growth Factor 23 and Cardiovascular Risk in Diabetes Patients-Cardiologists Be Aware.Metabolites. 2022 May 30;12(6):498. doi: 10.3390/metabo12060498. Metabolites. 2022. PMID: 35736431 Free PMC article.
References
-
- Burnett CH, Dent CE, Harper C, et al. Vitamin D-resistant rickets. Analysis of 24 pedigrees with hereditary and sporadic cases. Am J Med 1964; 36: 222–232. - PubMed
-
- Che H, Roux C, Etcheto A, et al. Impaired quality of life in adults with X-linked hypophosphatemia and skeletal symptoms. Eur J Endocrinol 2016; 174: 325–333. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
