A system that delivers an antioxidant to mitochondria for the treatment of drug-induced liver injury
- PMID: 37164988
- PMCID: PMC10172346
- DOI: 10.1038/s41598-023-33893-7
A system that delivers an antioxidant to mitochondria for the treatment of drug-induced liver injury
Abstract
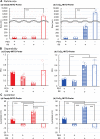
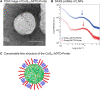
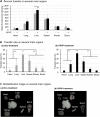
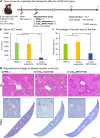
Mitochondria, a major source of reactive oxygen species (ROS), are intimately involved in the response to oxidative stress in the body. The production of excessive ROS affects the balance between oxidative responses and antioxidant defense mechanisms thus perturbing mitochondrial function eventually leading to tissue injury. Therefore, antioxidant therapies that target mitochondria can be used to treat such diseases and improve general health. This study reports on an attempt to establish a system for delivering an antioxidant molecule coenzyme Q10 (CoQ10) to mitochondria and the validation of its therapeutic efficacy in a model of acetaminophen (APAP) liver injury caused by oxidative stress in mitochondria. A CoQ10-MITO-Porter, a mitochondrial targeting lipid nanoparticle (LNP) containing encapsulated CoQ10, was prepared using a microfluidic device. It was essential to include polyethylene glycol (PEG) in the lipid composition of this LNP to ensure stability of the CoQ10, since it is relatively insoluble in water. Based on transmission electron microscope (TEM) observations and small angle X-ray scattering (SAXS) measurements, the CoQ10-MITO-Porter was estimated to be a 50 nm spherical particle without a regular layer structure. The use of the CoQ10-MITO-Porter improved liver function and reduced tissue injury, suggesting that it exerted a therapeutic effect on APAP liver injury.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Mitochondrial delivery of Coenzyme Q10 via systemic administration using a MITO-Porter prevents ischemia/reperfusion injury in the mouse liver.J Control Release. 2015 Sep 10;213:86-95. doi: 10.1016/j.jconrel.2015.06.037. Epub 2015 Jul 6. J Control Release. 2015. PMID: 26160304
-
Protective effects of coenzyme Q10 nanoparticles on dichlorvos-induced hepatotoxicity and mitochondrial/lysosomal injury.Environ Toxicol. 2018 Feb;33(2):167-177. doi: 10.1002/tox.22505. Epub 2017 Nov 16. Environ Toxicol. 2018. PMID: 29143438
-
The Use of a Microfluidic Device to Encapsulate a Poorly Water-Soluble Drug CoQ10 in Lipid Nanoparticles and an Attempt to Regulate Intracellular Trafficking to Reach Mitochondria.J Pharm Sci. 2019 Aug;108(8):2668-2676. doi: 10.1016/j.xphs.2019.04.001. Epub 2019 Apr 5. J Pharm Sci. 2019. PMID: 30959057
-
Oxidative stress during acetaminophen hepatotoxicity: Sources, pathophysiological role and therapeutic potential.Redox Biol. 2016 Dec;10:148-156. doi: 10.1016/j.redox.2016.10.001. Epub 2016 Oct 4. Redox Biol. 2016. PMID: 27744120 Free PMC article. Review.
-
Oxidant stress, mitochondria, and cell death mechanisms in drug-induced liver injury: lessons learned from acetaminophen hepatotoxicity.Drug Metab Rev. 2012 Feb;44(1):88-106. doi: 10.3109/03602532.2011.602688. Epub 2012 Jan 10. Drug Metab Rev. 2012. PMID: 22229890 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

