Safety of Janus Kinase inhibitors in Patients with Alopecia Areata: A Systematic Review
- PMID: 37138134
- PMCID: PMC10155665
- DOI: 10.1007/s40261-023-01260-z
Safety of Janus Kinase inhibitors in Patients with Alopecia Areata: A Systematic Review
Abstract
Background and objectives: Janus kinase (JAK) inhibitors are emerging as a therapeutic option for alopecia areata. The risk of potential adverse events is currently debated. In particular, several safety data for JAK inhibitors are extrapolated from a single study in elderly patients with rheumatoid arthritis treated with tofacitinib or adalimumab/etanercept as a comparator. The population of patients with alopecia areata is clinically and immunologically different from persons with rheumatoid arthritis and tumor necrosis factor (TNF) inhibitors are not effective in these patients. The objective of this systematic review was to analyze available data on the safety of various JAK inhibitors in patients with alopecia areata.
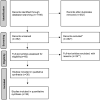
Methods: The systematic review was performed according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. A literature review was performed by searching PubMed, Scopus and EBSCO databases with the last search on March 13, 2023.
Results: In total, 36 studies were included. The frequency and odds ratio (OR) for most common adverse events versus placebo were: for baricitinib hypercholesterolemia (18.2% vs 10.5%, OR = 1.9) and headache (6.1% vs 5.1%, OR = 1.2), for brepocitinib elevated creatinine level (27.7% vs 4.3%, OR = 8.6) and acne (10.6% vs 4.3%, OR = 2.7), for ritlecitinib acne (10.4% vs 4.3%, OR = 2.6) and headache (12.5% vs 10.6%, OR = 1.2) and for deuruxolitinib headache (21.4% vs 9.1%, OR = 2.7) and acne (13.6% vs 4.5%, OR = 3.3). The respective numbers for upper respiratory infections were: baricitinib (7.3% vs 7.0%, OR = 1.0) and brepocitinib (23.4% vs 10.6%, OR = 2.6); for nasopharyngitis: ritlecitinib (12.5% vs 12.8%, OR = 1.0) and deuruxolitinib (14.6% vs 2.3%, OR = 7.3).
Conclusions: The most common side effects of JAK inhibitors in patients with alopecia areata were headache and acne. The OR for upper respiratory tract infections varied from over 7-fold increased to comparable to placebo. The risk of serious adverse events was not increased.
© 2023. The Author(s).
Conflict of interest statement
Małgorzata Papierzewska—no conflicts of interest. Anna Waśkiel-Burnat)—no conflicts of interest. Lidia Rudnicka—medical lectures for Leo Pharma, Novartis, UCB Pharma; participation in advisory board meetings of Leo Pharma, Novartis, Pfizer, Sanofi, and UCB Pharma; participation in advisory board of the EMA.
Figures
Similar articles
-
Comparative Efficacy and Safety of Janus Kinase Inhibitors Used in Alopecia Areata: A Systematic Review and Meta-analysis.Acta Derm Venereol. 2023 Jan 25;103:adv00855. doi: 10.2340/actadv.v103.4536. Acta Derm Venereol. 2023. PMID: 36695751 Free PMC article.
-
A phase 2a randomized, placebo-controlled study to evaluate the efficacy and safety of the oral Janus kinase inhibitors ritlecitinib and brepocitinib in alopecia areata: 24-week results.J Am Acad Dermatol. 2021 Aug;85(2):379-387. doi: 10.1016/j.jaad.2021.03.050. Epub 2021 Mar 20. J Am Acad Dermatol. 2021. PMID: 33757798 Clinical Trial.
-
Janus kinase inhibitors for alopecia areata.J Am Acad Dermatol. 2023 Aug;89(2S):S29-S32. doi: 10.1016/j.jaad.2023.05.049. J Am Acad Dermatol. 2023. PMID: 37591562 Review.
-
Systematic review of newer agents for the management of alopecia areata in adults: Janus kinase inhibitors, biologics and phosphodiesterase-4 inhibitors.J Eur Acad Dermatol Venereol. 2023 Apr;37(4):666-679. doi: 10.1111/jdv.18810. Epub 2022 Dec 22. J Eur Acad Dermatol Venereol. 2023. PMID: 36478475 Review.
-
LITFULOTM (Ritlecitinib) Capsules: A Janus Kinase 3 Inhibitor for the Treatment of Severe Alopecia Areata.Skinmed. 2023 Dec 5;21(6):434-438. eCollection 2023. Skinmed. 2023. PMID: 38051245
Cited by
-
Humanized CXCL12 antibody delays onset and modulates immune response in alopecia areata mice: insights from single-cell RNA sequencing.Front Immunol. 2024 Oct 17;15:1444777. doi: 10.3389/fimmu.2024.1444777. eCollection 2024. Front Immunol. 2024. PMID: 39483478 Free PMC article.
-
Efficacy and Safety of Topical Tofacitinib for the Treatment of Alopecia Areata.Indian Dermatol Online J. 2024 Jun 26;15(4):624-629. doi: 10.4103/idoj.idoj_535_23. eCollection 2024 Jul-Aug. Indian Dermatol Online J. 2024. PMID: 39050046 Free PMC article.
References
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical

