Antihepatoma peptide, scolopentide, derived from the centipede scolopendra subspinipes mutilans
- PMID: 37032730
- PMCID: PMC10080696
- DOI: 10.3748/wjg.v29.i12.1875
Antihepatoma peptide, scolopentide, derived from the centipede scolopendra subspinipes mutilans
Abstract
Background: Centipedes have been used to treat tumors for hundreds of years in China. However, current studies focus on antimicrobial and anticoagulation agents rather than tumors. The molecular identities of antihepatoma bioactive components in centipedes have not yet been extensively investigated. It is a challenge to isolate and characterize the effective components of centipedes due to limited peptide purification technologies for animal-derived medicines.
Aim: To purify, characterize, and synthesize the bioactive components with the strongest antihepatoma activity from centipedes and determine the antihepatoma mechanism.
Methods: An antihepatoma peptide (scolopentide) was isolated and identified from the centipede scolopendra subspinipes mutilans using a combination of enzymatic hydrolysis, a Sephadex G-25 column, and two steps of high-performance liquid chromatography (HPLC). Additionally, the CCK8 assay was used to select the extracted fraction with the strongest antihepatoma activity. The molecular weight of the extracted scolopentide was characterized by quadrupole time of flight mass spectrometry (QTOF MS), and the sequence was matched by using the Mascot search engine. Based on the sequence and molecular weight, scolopentide was synthesized using solid-phase peptide synthesis methods. The synthetic scolopentide was confirmed by MS and HPLC. The antineoplastic effect of extracted scolopentide was confirmed by CCK8 assay and morphological changes again in vitro. The antihepatoma effect of synthetic scolopentide was assessed by the CCK8 assay and Hoechst staining in vitro and tumor volume and tumor weight in vivo. In the tumor xenograft experiments, qualified model mice (male 5-week-old BALB/c nude mice) were randomly divided into 2 groups (n = 6): The scolopentide group (0.15 mL/d, via intraperitoneal injection of synthetic scolopentide, 500 mg/kg/d) and the vehicle group (0.15 mL/d, via intraperitoneal injection of normal saline). The mice were euthanized by cervical dislocation after 14 d of continuous treatment. Mechanistically, flow cytometry was conducted to evaluate the apoptosis rate of HepG2 cells after treatment with extracted scolopentide in vitro. A Hoechst staining assay was also used to observe apoptosis in HepG2 cells after treatment with synthetic scolopentide in vitro. CCK8 assays and morphological changes were used to compare the cytotoxicity of synthetic scolopentide to liver cancer cells and normal liver cells in vitro. Molecular docking was performed to clarify whether scolopentide tightly bound to death receptor 4 (DR4) and DR5. qRT-PCR was used to measure the mRNA expression of DR4, DR5, fas-associated death domain protein (FADD), Caspase-8, Caspase-3, cytochrome c (Cyto-C), B-cell lymphoma-2 (Bcl-2), Bcl-2-associated X protein (Bax), x-chromosome linked inhibitor-of-apoptosis protein and Cellular fas-associated death domain-like interleukin-1β converting enzyme inhibitory protein in hepatocarcinoma subcutaneous xenograft tumors from mice. Western blot assays were used to measure the protein expression of DR4, DR5, FADD, Caspase-8, Caspase-3, and Cyto-C in the tumor tissues. The reactive oxygen species (ROS) of tumor tissues were tested.
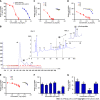
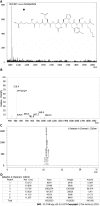
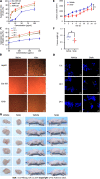
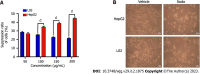
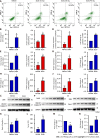
Results: In the process of purification, characterization and synthesis of scolopentide, the optimal enzymatic hydrolysis conditions (extract ratio: 5.86%, IC50: 0.310 mg/mL) were as follows: Trypsin at 0.1 g (300 U/g, centipede-trypsin ratio of 20:1), enzymolysis temperature of 46 °C, and enzymolysis time of 4 h, which was superior to freeze-thawing with liquid nitrogen (IC50: 3.07 mg/mL). A peptide with the strongest antihepatoma activity (scolopentide) was further purified through a Sephadex G-25 column (obtained A2) and two steps of HPLC (obtained B5 and C3). The molecular weight of the extracted scolopentide was 1018.997 Da, and the peptide sequence was RAQNHYCK, as characterized by QTOF MS and Mascot. Scolopentide was synthesized in vitro with a qualified molecular weight (1018.8 Da) and purity (98.014%), which was characterized by MS and HPLC. Extracted scolopentide still had an antineoplastic effect in vitro, which inhibited the proliferation of Eca-109 (IC50: 76.27 μg/mL), HepG2 (IC50: 22.06 μg/mL), and A549 (IC50: 35.13 μg/mL) cells, especially HepG2 cells. Synthetic scolopentide inhibited the proliferation of HepG2 cells (treated 6, 12, and 24 h) in a concentration-dependent manner in vitro, and the inhibitory effects were the strongest at 12 h (IC50: 208.11 μg/mL). Synthetic scolopentide also inhibited the tumor volume (Vehicle vs Scolopentide, P = 0.0003) and weight (Vehicle vs Scolopentide, P = 0.0022) in the tumor xenograft experiment. Mechanistically, flow cytometry suggested that the apoptosis ratios of HepG2 cells after treatment with extracted scolopentide were 5.01% (0 μg/mL), 12.13% (10 μg/mL), 16.52% (20 μg/mL), and 23.20% (40 μg/mL). Hoechst staining revealed apoptosis in HepG2 cells after treatment with synthetic scolopentide in vitro. The CCK8 assay and morphological changes indicated that synthetic scolopentide was cytotoxic and was significantly stronger in HepG2 cells than in L02 cells. Molecular docking suggested that scolopentide tightly bound to DR4 and DR5, and the binding free energies were-10.4 kcal/mol and-7.1 kcal/mol, respectively. In subcutaneous xenograft tumors from mice, quantitative real-time polymerase chain reaction and western blotting suggested that scolopentide activated DR4 and DR5 and induced apoptosis in SMMC-7721 Liver cancer cells by promoting the expression of FADD, caspase-8 and caspase-3 through a mitochondria-independent pathway.
Conclusion: Scolopentide, an antihepatoma peptide purified from centipedes, may inspire new antihepatoma agents. Scolopentide activates DR4 and DR5 and induces apoptosis in liver cancer cells through a mitochondria-independent pathway.
Keywords: Antihepatom peptide; Centipede; Death receptor 4; Death receptor 5; Hepatocellular carcinoma; Scolopendra.
©The Author(s) 2023. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: The authors declare that they have no competing interests.
Figures
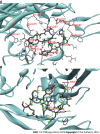

Similar articles
-
Total alkaloids of Rubus aleaefolius Poir inhibit hepatocellular carcinoma growth in vivo and in vitro via activation of mitochondrial-dependent apoptosis.Int J Oncol. 2013 Mar;42(3):971-8. doi: 10.3892/ijo.2013.1779. Epub 2013 Jan 18. Int J Oncol. 2013. PMID: 23338043
-
Alpinia oxyphylla oil induces apoptosis of hepatocellular carcinoma cells via PI3K/Akt pathway in vitro and in vivo.Biomed Pharmacother. 2019 Jan;109:2365-2374. doi: 10.1016/j.biopha.2018.11.124. Epub 2018 Nov 30. Biomed Pharmacother. 2019. PMID: 30551496
-
Antitumor Efficacy of Huqizhengxiao (HQZX) Decoction Based on Inhibition of Telomerase Activity in Nude Mice of Hepatocarcinoma Xenograft.Integr Cancer Ther. 2018 Dec;17(4):1216-1224. doi: 10.1177/1534735418785999. Epub 2018 Jul 6. Integr Cancer Ther. 2018. PMID: 29978739 Free PMC article.
-
Synthesis, biological activities and mechanistic studies of C20-ketone pachysandra alkaloids as anti-hepatocellular carcinoma agents.Mol Divers. 2024 Aug 19. doi: 10.1007/s11030-024-10961-2. Online ahead of print. Mol Divers. 2024. PMID: 39158620 Review.
-
Regulation of anoikis by extrinsic death receptor pathways.Cell Commun Signal. 2023 Sep 4;21(1):227. doi: 10.1186/s12964-023-01247-5. Cell Commun Signal. 2023. PMID: 37667281 Free PMC article. Review.
Cited by
-
Role of ACSL4 in modulating farnesoid X receptor expression and M2 macrophage polarization in HBV-induced hepatocellular carcinoma.MedComm (2020). 2024 Sep 12;5(9):e706. doi: 10.1002/mco2.706. eCollection 2024 Sep. MedComm (2020). 2024. PMID: 39268355 Free PMC article.
-
Scorpiones, Scolopendra and Gekko Inhibit Lung Cancer Growth and Metastasis by Ameliorating Hypoxic Tumor Microenvironment via PI3K/AKT/mTOR/HIF-1α Signaling Pathway.Chin J Integr Med. 2024 Sep;30(9):799-808. doi: 10.1007/s11655-024-3803-8. Epub 2024 Jun 8. Chin J Integr Med. 2024. PMID: 38850481
References
-
- Commission NP. Pharmacopoeia of the People’s Republic of China. 2020. [cited 3 September 2022]. Available from: https://www.semanticscholar.org/paper/Pharmacopoeia-of-the-People%27s-Re... .
-
- Xu XL, Wang CM, Geng D, Zhang L, Hu B. [Effects of centipede extracts on normal mouse and S180, H22 bearing mouse] Zhong Yao Cai. 2010;33:499–503. - PubMed
-
- Ding D, Guo YR, Wu RL, Qi WY, Xu HM. Two new isoquinoline alkaloids from Scolopendra subspinipes mutilans induce cell cycle arrest and apoptosis in human glioma cancer U87 cells. Fitoterapia. 2016;110:103–109. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

