Retraction of Clinical Trials about the SARS-CoV-2 Infection: An Unaddressed Problem and Its Possible Impact on Coronavirus Disease (COVID)-19 Treatment
- PMID: 36767202
- PMCID: PMC9914919
- DOI: 10.3390/ijerph20031835
Retraction of Clinical Trials about the SARS-CoV-2 Infection: An Unaddressed Problem and Its Possible Impact on Coronavirus Disease (COVID)-19 Treatment
Abstract
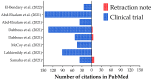
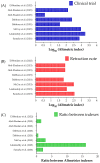
We are presenting an overview of the retracted clinical trials about the Coronavirus Disease (COVID)-19 published in PubMed using the descriptors ((COVID-19 OR SARS-CoV-2) AND (Clinical Trial)). We collected the information for i) the first author's country; ii) the journal name where the study was published; iii) the impact factor of the journal; iv) the main objective of the study; v) methods including population, intervention, study design, and outcomes; and vi) results and conclusions. We collected complete information from the retraction notes published by the journals and the number of publications/retractions related to non-COVID-19 clinical trials published simultaneously. We also included the Altmetric index for the clinical trials and the retraction notes about COVID-19 to compare the accessibility to both studies' indexes. The retraction of clinical trials occurred in four countries (one in Lebanon, one in India, one in Brazil, and five in Egypt) and six journals (one in Viruses, one in Archives of Virology, one in Expert Review of Anti-infective Therapy, one in Frontiers in Medicine, two in Scientific Reports, and two in The American Journal of Tropical Medicine and Hygiene). Eight drugs were tested (Ivermectin, Vitamin D, Proxalutamide, Hydroxychloroquine, Remdesevir, Favipiravir, and Sofosbuvir + Daclatasvir) in the studies. One of the retractions was suggested by the authors due to an error in the statistical analysis, which compromised their results and conclusions. Also, the methods, mainly the allocation, were not well conducted in the two studies, and the studies were retracted. In addition, the studies performed by Dabbous et al. presented several issues, mainly including several raw datasets that did not prove their findings. Moreover, two studies were retracted due to data overlap and copying. Significant concerns were raised about the integrity of the data and reported results in another article. We identified a higher Altmetric index for the original studies, proving that the retracted studies were accessed more than the retraction notes. Interestingly, the impact of the original articles is much higher than their retraction notes. The different Altmetric indexes show that possibly people who read those retracted articles are not reading their retraction notes and are unaware of the erroneous information they share. COVID-19- related clinical trials were ~two-time times more retracted than the other clinical trials performed during the same time.
Keywords: SARS-CoV-2; clinical trial; mortality; pandemic; science.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
Retracted articles in the obstetrics literature: lessons from the past to change the future.Am J Obstet Gynecol MFM. 2020 Nov;2(4):100201. doi: 10.1016/j.ajogmf.2020.100201. Epub 2020 Aug 19. Am J Obstet Gynecol MFM. 2020. PMID: 33345918
-
Inconsistent and incomplete retraction of published research: A cross-sectional study on Covid-19 retractions and recommendations to mitigate risks for research, policy and practice.PLoS One. 2021 Oct 27;16(10):e0258935. doi: 10.1371/journal.pone.0258935. eCollection 2021. PLoS One. 2021. PMID: 34705841 Free PMC article.
-
Reasons for article retraction in anesthesiology: a comprehensive analysis.Can J Anaesth. 2020 Jan;67(1):57-63. doi: 10.1007/s12630-019-01508-3. Epub 2019 Oct 15. Can J Anaesth. 2020. PMID: 31617069 Review. English.
-
Anesthesiology Articles Published in 2020: A Review and Characterization of COVID-19 Versus Non-COVID-19 Publications in Top Anesthesiology Journals.Cureus. 2022 Apr 8;14(4):e23943. doi: 10.7759/cureus.23943. eCollection 2022 Apr. Cureus. 2022. PMID: 35547422 Free PMC article. Review.
Cited by
-
Efficacy of Ivermectin, Chloroquine/Hydroxychloroquine, and Azithromycin in Managing COVID-19: A Systematic Review of Phase III Clinical Trials.Biomedicines. 2024 Sep 27;12(10):2206. doi: 10.3390/biomedicines12102206. Biomedicines. 2024. PMID: 39457519 Free PMC article. Review.
-
Phase 4 clinical trials in the era of the Coronavirus Disease (COVID-19) pandemic and their importance to optimize the COVID-19 vaccination.Hum Vaccin Immunother. 2023 Aug 1;19(2):2234784. doi: 10.1080/21645515.2023.2234784. Hum Vaccin Immunother. 2023. PMID: 37449956 Free PMC article. Clinical Trial.
-
Assessment of the development of social skills among undergraduate medical students through participation in a socio-educational project during the coronavirus disease (COVID)-19 pandemic: a Brazilian pilot study.BMC Med Educ. 2024 Oct 23;24(1):1197. doi: 10.1186/s12909-024-06188-2. BMC Med Educ. 2024. PMID: 39443877 Free PMC article.
-
Putting your trust in ICU clinical trials: the journal's role.Intensive Care Med. 2023 Dec;49(12):1520-1522. doi: 10.1007/s00134-023-07235-0. Epub 2023 Oct 16. Intensive Care Med. 2023. PMID: 37843571 No abstract available.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

