SigB modulates expression of novel SigB regulon members via Bc1009 in non-stressed and heat-stressed cells revealing its alternative roles in Bacillus cereus
- PMID: 36759782
- PMCID: PMC9912610
- DOI: 10.1186/s12866-023-02783-3
SigB modulates expression of novel SigB regulon members via Bc1009 in non-stressed and heat-stressed cells revealing its alternative roles in Bacillus cereus
Abstract
Background: The Bacillus cereus Sigma B (SigB) dependent general stress response is activated via the two-component RsbKY system, which involves a phosphate transfer from RsbK to RsbY. It has been hypothesized that the Hpr-like phosphocarrier protein (Bc1009) encoded by bc1009 in the SigB gene cluster may play a role in this transfer, thereby acting as a regulator of SigB activation. Alternatively, Bc1009 may be involved in the activation of a subset of SigB regulon members.
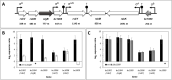
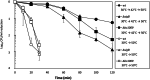
Results: We first investigated the potential role of bc1009 to act as a SigB regulator but ruled out this possibility as the deletion of bc1009 did not affect the expression of sigB and other SigB gene cluster members. The SigB-dependent functions of Bc1009 were further examined in B. cereus ATCC14579 via comparative proteome profiling (backed up by transcriptomics) of wt, Δbc1009 and ΔsigB deletion mutants under heat stress at 42 °C. This revealed 284 proteins displaying SigB-dependent alterations in protein expression levels in heat-stressed cells, including a subgroup of 138 proteins for which alterations were also Bc1009-dependent. Next to proteins with roles in stress defense, newly identified SigB and Bc1009-dependent proteins have roles in cell motility, signal transduction, transcription, cell wall biogenesis, and amino acid transport and metabolism. Analysis of lethal stress survival at 50 °C after pre-adaptation at 42 °C showed intermediate survival efficacy of Δbc1009 cells, highest survival of wt, and lowest survival of ΔsigB cells, respectively. Additional comparative proteome analysis of non-stressed wt and mutant cells at 30 °C revealed 96 proteins with SigB and Bc1009-dependent differences in levels: 51 were also identified under heat stress, and 45 showed significant differential expression at 30 °C. This includes proteins with roles in carbohydrate/ion transport and metabolism. Overlapping functions at 30 °C and 42 °C included proteins involved in motility, and ΔsigB and Δbc1009 cells showed reduced motility compared to wt cells in swimming assays at both temperatures.
Conclusion: Our results extend the B. cereus SigB regulon to > 300 members, with a novel role of SigB-dependent Bc1009 in the activation of a subregulon of > 180 members, conceivably via interactions with other transcriptional regulatory networks.
Keywords: Adaptive general stress response; Amino acid metabolisms; Metabolic crosstalk; Motility; Phosphocarrier protein; SigB baseline function; SigB subregulon; Sporulation; Virulence.
© 2023. The Author(s).
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures


Similar articles
-
Prediction and validation of novel SigB regulon members in Bacillus subtilis and regulon structure comparison to Bacillales members.BMC Microbiol. 2023 Jan 18;23(1):17. doi: 10.1186/s12866-022-02700-0. BMC Microbiol. 2023. PMID: 36653740 Free PMC article.
-
The response of Bacillus licheniformis to heat and ethanol stress and the role of the SigB regulon.Proteomics. 2013 Jul;13(14):2140-61. doi: 10.1002/pmic.201200297. Proteomics. 2013. PMID: 23592518
-
Chill induction of the SigB-dependent general stress response in Bacillus subtilis and its contribution to low-temperature adaptation.J Bacteriol. 2003 Aug;185(15):4305-14. doi: 10.1128/JB.185.15.4305-4314.2003. J Bacteriol. 2003. PMID: 12867438 Free PMC article.
-
Non-specific, general and multiple stress resistance of growth-restricted Bacillus subtilis cells by the expression of the sigmaB regulon.Mol Microbiol. 1998 Sep;29(5):1129-36. doi: 10.1046/j.1365-2958.1998.00977.x. Mol Microbiol. 1998. PMID: 9767581 Review.
-
The Stress-Responsive Alternative Sigma Factor SigB of Bacillus subtilis and Its Relatives: An Old Friend With New Functions.Front Microbiol. 2020 Sep 15;11:1761. doi: 10.3389/fmicb.2020.01761. eCollection 2020. Front Microbiol. 2020. PMID: 33042030 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

