Cushing's syndrome: a combined treatment with etomidate and osilodrostat in severe life-threatening hypercortisolemia
- PMID: 36129663
- PMCID: PMC9712315
- DOI: 10.1007/s42000-022-00397-4
Cushing's syndrome: a combined treatment with etomidate and osilodrostat in severe life-threatening hypercortisolemia
Abstract
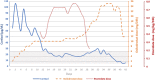
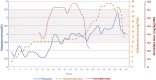
Endogenous Cushing's syndrome (CS) is associated with increased morbidity and mortality. Early diagnosis and initiation of therapy are essential, but effective treatment remains a challenge. In a long-term follow-up, biochemical control of hypercortisolemia, especially when severe, is difficult to achieve. Life-threatening hypercortisolemia is difficult to control due to the limitations of pharmacotherapy, including its side effects, and may require etomidate infusion in the intensive care unit (ICU) to rapidly lower cortisol levels. The effectiveness of hypercortisolemia management can be increased by a dual blockade of cortisol production. We report the efficacy, safety, and tolerability of combined therapy with two steroidogenesis inhibitors, etomidate, and osilodrostat, in a 32-year-old woman diagnosed with severe ACTH-dependent hypercortisolemia, subsequently maintaining a stable level of cortisol with osilodrostat monotherapy. This approach enabled achievement of relatively rapid control of the hypercortisolemia while using an etomidate infusion and concomitant increasing doses of oral osilodrostat applying a "titrations strategy." Our experience shows that it is worth taking advantage of the synergistic anticortisolic action of etomidate with osilodrostat.
Keywords: Cortisol; Cushing’s syndrome; Etomidate; Osilodrostat; Severe hypercortisolemia.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Updates in adrenal steroidogenesis inhibitors for Cushing's syndrome - A practical guide.Best Pract Res Clin Endocrinol Metab. 2021 Jan;35(1):101490. doi: 10.1016/j.beem.2021.101490. Epub 2021 Feb 6. Best Pract Res Clin Endocrinol Metab. 2021. PMID: 33707082 Review.
-
A novel use of etomidate for intentional adrenal suppression to control severe hypercortisolemia in childhood.Pediatr Crit Care Med. 2009 May;10(3):e37-40. doi: 10.1097/PCC.0b013e318198b096. Pediatr Crit Care Med. 2009. PMID: 19433940
-
Continuous Etomidate for the Management of Cushing's Syndrome Complicated by Pulmonary Nocardiosis.J Pharm Pract. 2022 Dec;35(6):1057-1059. doi: 10.1177/08971900211017487. Epub 2021 May 31. J Pharm Pract. 2022. PMID: 34056961
-
Etomidate infusion at low doses is an effective and safe treatment for severe Cushing's syndrome outside intensive care.Eur J Endocrinol. 2020 Aug;183(2):161-167. doi: 10.1530/EJE-20-0380. Eur J Endocrinol. 2020. PMID: 32449698
-
Pharmacological management of severe Cushing's syndrome: the role of etomidate.Ther Adv Endocrinol Metab. 2022 Feb 14;13:20420188211058583. doi: 10.1177/20420188211058583. eCollection 2022. Ther Adv Endocrinol Metab. 2022. PMID: 35186251 Free PMC article. Review.
Cited by
-
Etomidate-induced hypokalemia in electronic cigarette users: two case reports and literature review.Front Endocrinol (Lausanne). 2024 May 30;15:1321610. doi: 10.3389/fendo.2024.1321610. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 38872965 Free PMC article. Review.
-
Genetics of hyperaldosteronism and a wealth of new information on topics ranging from MEN1 to Cushing's disease and metabolic syndrome.Hormones (Athens). 2022 Dec;21(4):521-523. doi: 10.1007/s42000-022-00418-2. Hormones (Athens). 2022. PMID: 36380266 No abstract available.
-
Osilodrostat: A Novel Potent Inhibitor of 11-Beta-Hydroxylase for the Treatment of Cushing's Syndrome.touchREV Endocrinol. 2024 Apr;20(1):43-51. doi: 10.17925/EE.2024.20.1.8. Epub 2023 Dec 11. touchREV Endocrinol. 2024. PMID: 38812665 Free PMC article. Review.
-
Ectopic Cushing Syndrome Secondary to Diffuse Idiopathic Neuroendocrine Cell Hyperplasia-A Report of 2 Cases.JCEM Case Rep. 2024 Aug 6;2(8):luae128. doi: 10.1210/jcemcr/luae128. eCollection 2024 Aug. JCEM Case Rep. 2024. PMID: 39108602 Free PMC article.
-
Cushing's Disease: Long-Term Effectiveness and Safety of Osilodrostat in a Polish Group of Patients with Persistent Hypercortisolemia in the Experience of a Single Center.Biomedicines. 2023 Dec 6;11(12):3227. doi: 10.3390/biomedicines11123227. Biomedicines. 2023. PMID: 38137448 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

