Science-Driven Nutritional Interventions for the Prevention and Treatment of Cancer
- PMID: 35997502
- PMCID: PMC10749912
- DOI: 10.1158/2159-8290.CD-22-0504
Science-Driven Nutritional Interventions for the Prevention and Treatment of Cancer
Abstract
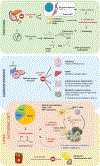
In population studies, dietary patterns clearly influence the development, progression, and therapeutic response of cancers. Nonetheless, interventional dietary trials have had relatively little impact on the prevention and treatment of malignant disease. Standardization of nutritional interventions combined with high-level mode-of-action studies holds the promise of identifying specific entities and pathways endowed with antineoplastic properties. Here, we critically review the effects of caloric restriction and more specific interventions on macro- and micronutrients in preclinical models as well as in clinical studies. We place special emphasis on the prospect of using defined nutrition-relevant molecules to enhance the efficacy of established anticancer treatments.
Significance: The avoidance of intrinsically hypercaloric and toxic diets contributes to the prevention and cure of cancer. In addition, specific diet-induced molecules such as ketone bodies and micronutrients, including specific vitamins, have drug-like effects that are clearly demonstrable in preclinical models, mostly in the context of immunotherapies. Multiple trials are underway to determine the clinical utility of such molecules.
©2022 American Association for Cancer Research.
Figures

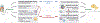
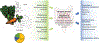
Similar articles
-
Micronutrients Importance in Cancer Prevention-Vitamins.Cancer Treat Res. 2024;191:119-144. doi: 10.1007/978-3-031-55622-7_5. Cancer Treat Res. 2024. PMID: 39133406 Review.
-
The role of cellular micronutrient analysis, nutraceuticals, vitamins, antioxidants and minerals in the prevention and treatment of hypertension and cardiovascular disease.Ther Adv Cardiovasc Dis. 2010 Jun;4(3):165-83. doi: 10.1177/1753944710368205. Epub 2010 Apr 16. Ther Adv Cardiovasc Dis. 2010. PMID: 20400494 Review.
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
Recommendations for Nutritional Supplementation in Pediatric Oncology: A Compilation of the Facts.Nutrients. 2023 Jul 21;15(14):3239. doi: 10.3390/nu15143239. Nutrients. 2023. PMID: 37513658 Free PMC article. Review.
-
Strengthening the immunity of the Swiss population with micronutrients: A narrative review and call for action.Clin Nutr ESPEN. 2021 Jun;43:39-48. doi: 10.1016/j.clnesp.2021.03.012. Epub 2021 Mar 24. Clin Nutr ESPEN. 2021. PMID: 34024545 Free PMC article. Review.
Cited by
-
Developing the next-generation cancer research workforce in the National Institutes of Health Intramural Research Program.J Natl Cancer Inst. 2024 May 8;116(5):637-641. doi: 10.1093/jnci/djae017. J Natl Cancer Inst. 2024. PMID: 38273662 Free PMC article.
-
The increase in cell death rates in caloric restricted cells of the yeast helicase mutant rrm3 is Sir complex dependent.Sci Rep. 2023 Oct 19;13(1):17832. doi: 10.1038/s41598-023-45125-z. Sci Rep. 2023. PMID: 37857740 Free PMC article.
-
Tumor initiation and early tumorigenesis: molecular mechanisms and interventional targets.Signal Transduct Target Ther. 2024 Jun 19;9(1):149. doi: 10.1038/s41392-024-01848-7. Signal Transduct Target Ther. 2024. PMID: 38890350 Free PMC article. Review.
-
An Exogenous Ketone Ester Slows Tumor Progression in Murine Breast and Renal Cancer Models.Cancers (Basel). 2024 Oct 4;16(19):3390. doi: 10.3390/cancers16193390. Cancers (Basel). 2024. PMID: 39410010 Free PMC article.
-
Complementarity between Microbiome and Immunity May Account for the Potentiating Effect of Quercetin on the Antitumor Action of Cyclophosphamide in a Triple-Negative Breast Cancer Model.Pharmaceuticals (Basel). 2023 Oct 6;16(10):1422. doi: 10.3390/ph16101422. Pharmaceuticals (Basel). 2023. PMID: 37895893 Free PMC article.
References
-
- Hanahan D. Hallmarks of cancer: new dimensions. Cancer Discov 2022;12:31–46. - PubMed
-
- Lopez-Otin C, Kroemer G. Hallmarks of health. Cell 2021;184:33–63. - PubMed
-
- Derosa L, Routy B, Desilets A, Daillere R, Terrisse S, Kroemer G, et al. Microbiota-centered interventions: the next breakthrough in immuno-oncology? Cancer Discov 2021;11:2396–412. - PubMed
-
- Sharma P, Siddiqui BA, Anandhan S, Yadav SS, Subudhi SK, Gao J, et al. The next decade of immune checkpoint therapy. Cancer Discov 2021;11:838–57. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

