Comparison Between Dichloroacetate and Phenylbutyrate Treatment for Pyruvate Dehydrogenase Deficiency
- PMID: 35996497
- PMCID: PMC9302545
- DOI: 10.3389/bjbs.2022.10382
Comparison Between Dichloroacetate and Phenylbutyrate Treatment for Pyruvate Dehydrogenase Deficiency
Abstract
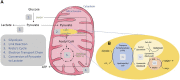
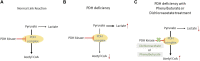
Pyruvate dehydrogenase (PDH) deficiency is caused by a number of pathogenic variants and the most common are found in the PDHA1 gene. The PDHA1 gene encodes one of the subunits of the PDH enzyme found in a carbohydrate metabolism pathway involved in energy production. Pathogenic variants of PDHA1 gene usually impact the α-subunit of PDH causing energy reduction. It potentially leads to increased mortality in sufferers. Potential treatments for this disease include dichloroacetate and phenylbutyrate, previously used for other diseases such as cancer and maple syrup urine disease. However, not much is known about their efficacy in treating PDH deficiency. Effective treatment for PDH deficiency is crucial as carbohydrate is needed in a healthy diet and rice is the staple food for a large portion of the Asian population. This review analysed the efficacy of dichloroacetate and phenylbutyrate as potential treatments for PDH deficiency caused by PDHA1 pathogenic variants. Based on the findings of this review, dichloroacetate will have an effect on most PDHA1 pathogenic variant and can act as a temporary treatment to reduce the lactic acidosis, a common symptom of PDH deficiency. Phenylbutyrate can only be used on patients with certain pathogenic variants (p.P221L, p.R234G, p.G249R, p.R349C, p.R349H) on the PDH protein. It is hoped that the review would provide an insight into these treatments and improve the quality of lives for patients with PDH deficiency.
Keywords: E1a; PDHA1; inborn error of metabolism; lactic acidosis; mitochondrial disease.
Copyright © 2022 Karissa, Simpson, Dawson, Low, Tay, Nordin, Zain, Lee and Pung.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Mild phenotype in a male with pyruvate dehydrogenase complex deficiency associated with novel hemizygous in-frame duplication of the E1α subunit gene (PDHA1).Neuropediatrics. 2014 Feb;45(1):56-60. doi: 10.1055/s-0033-1341601. Epub 2013 Apr 9. Neuropediatrics. 2014. PMID: 23572181
-
Neonatal pyruvate dehydrogenase deficiency due to a R302H mutation in the PDHA1 gene: MRI findings.Pediatr Radiol. 2008 May;38(5):559-62. doi: 10.1007/s00247-007-0721-9. Epub 2008 Jan 16. Pediatr Radiol. 2008. PMID: 18197404
-
Pyruvate dehydrogenase complex deficiency and its relationship with epilepsy frequency--An overview.Epilepsy Res. 2015 Oct;116:40-52. doi: 10.1016/j.eplepsyres.2015.07.002. Epub 2015 Jul 8. Epilepsy Res. 2015. PMID: 26354166 Review.
-
A cognitively normal PDH-deficient 18-year-old man carrying the R263G mutation in the PDHA1 gene.J Inherit Metab Dis. 2009 Dec;32 Suppl 1. doi: 10.1007/s10545-009-1101-4. Epub 2009 Jul 29. J Inherit Metab Dis. 2009. PMID: 19639391
-
Mutations in the X-linked pyruvate dehydrogenase (E1) alpha subunit gene (PDHA1) in patients with a pyruvate dehydrogenase complex deficiency.Hum Mutat. 2000;15(3):209-19. doi: 10.1002/(SICI)1098-1004(200003)15:3<209::AID-HUMU1>3.0.CO;2-K. Hum Mutat. 2000. PMID: 10679936 Review.
Cited by
-
RNF4 mediated degradation of PDHA1 promotes colorectal cancer metabolism and metastasis.NPJ Precis Oncol. 2024 Nov 9;8(1):258. doi: 10.1038/s41698-024-00724-5. NPJ Precis Oncol. 2024. PMID: 39521913 Free PMC article.
-
Phenylbutyrate and Dichloroacetate Enhance the Liquid-Stored Boar Sperm Quality via PDK1 and PDK3.Int J Mol Sci. 2023 Dec 4;24(23):17091. doi: 10.3390/ijms242317091. Int J Mol Sci. 2023. PMID: 38069413 Free PMC article.
-
Inborn Errors of Metabolism with Ataxia: Current and Future Treatment Options.Cells. 2023 Sep 19;12(18):2314. doi: 10.3390/cells12182314. Cells. 2023. PMID: 37759536 Free PMC article. Review.
References
-
- Garg U, Smith LD. Introduction to Laboratory Diagnosis and Biomarkers in Inborn Error of Metabolism. Biomarkers Inborn Errors Metab (2017):1–24. 10.1016/b978-0-12-802896-4.00001-8 - DOI
-
- Vasam G, Reid K, Burelle Y, Menzies KJ. Nutritional Regulation of Mitochondrial Function. Mitochondria Obes Type 2 Diabetes (2019) 93–126. 10.1016/B978-0-12-811752-1.00004-3 - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

