Ultraviolet-C Irradiation, Heat, and Storage as Potential Methods of Inactivating SARS-CoV-2 and Bacterial Pathogens on Filtering Facepiece Respirators
- PMID: 35056031
- PMCID: PMC8780977
- DOI: 10.3390/pathogens11010083
Ultraviolet-C Irradiation, Heat, and Storage as Potential Methods of Inactivating SARS-CoV-2 and Bacterial Pathogens on Filtering Facepiece Respirators
Abstract
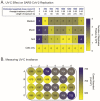
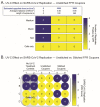
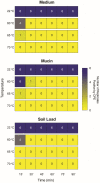
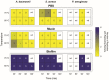
The arrival of SARS-CoV-2 to Aotearoa/New Zealand in February 2020 triggered a massive response at multiple levels. Procurement and sustainability of medical supplies to hospitals and clinics during the then upcoming COVID-19 pandemic was one of the top priorities. Continuing access to new personal protective equipment (PPE) was not guaranteed; thus, disinfecting and reusing PPE was considered as a potential alternative. Here, we describe part of a local program intended to test and implement a system to disinfect PPE for potential reuse in New Zealand. We used filtering facepiece respirator (FFR) coupons inoculated with SARS-CoV-2 or clinically relevant multidrug-resistant pathogens (Acinetobacter baumannii Ab5075, methicillin-resistant Staphylococcus aureus USA300 LAC and cystic-fibrosis isolate Pseudomonas aeruginosa LESB58), to evaluate the potential use of ultraviolet-C germicidal irradiation (UV-C) or dry heat treatment to disinfect PPE. An applied UV-C dose of 1000 mJ/cm2 was sufficient to completely inactivate high doses of SARS-CoV-2; however, irregularities in the FFR coupons hindered the efficacy of UV-C to fully inactivate the virus, even at higher UV-C doses (2000 mJ/cm2). Conversely, incubating contaminated FFR coupons at 65 °C for 30 min or 70 °C for 15 min, was sufficient to block SARS-CoV-2 replication, even in the presence of mucin or a soil load (mimicking salivary or respiratory secretions, respectively). Dry heat (90 min at 75 °C to 80 °C) effectively killed 106 planktonic bacteria; however, even extending the incubation time up to two hours at 80 °C did not completely kill bacteria when grown in colony biofilms. Importantly, we also showed that FFR material can harbor replication-competent SARS-CoV-2 for up to 35 days at room temperature in the presence of a soil load. We are currently using these findings to optimize and establish a robust process for decontaminating, reusing, and reducing wastage of PPE in New Zealand.
Keywords: COVID-19; New Zealand; PPE; SARS-CoV-2; UV-C; bacteria; disinfection; personal protective equipment.
Conflict of interest statement
The authors declare no conflict of interests, including no further patents, products in development or marketed products. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
Similar articles
-
Efficacy of Dry Heat Treatment against Clostridioides difficile Spores and Mycobacterium tuberculosis on Filtering Facepiece Respirators.Pathogens. 2022 Aug 2;11(8):871. doi: 10.3390/pathogens11080871. Pathogens. 2022. PMID: 36014991 Free PMC article.
-
Filtering Facepiece Respirator (N95 Respirator) Reprocessing: A Systematic Review.JAMA. 2021 Apr 6;325(13):1296-1317. doi: 10.1001/jama.2021.2531. JAMA. 2021. PMID: 33656543
-
The use of germicidal ultraviolet light, vaporised hydrogen peroxide and dry heat to decontaminate face masks and filtering respirators contaminated with an infectious norovirus.Infect Prev Pract. 2021 Mar;3(1):100111. doi: 10.1016/j.infpip.2020.100111. Epub 2020 Dec 28. Infect Prev Pract. 2021. PMID: 34316573 Free PMC article.
-
Rapid Review of SARS-CoV-1 and SARS-CoV-2 Viability, Susceptibility to Treatment, and the Disinfection and Reuse of PPE, Particularly Filtering Facepiece Respirators.Int J Environ Res Public Health. 2020 Aug 22;17(17):6117. doi: 10.3390/ijerph17176117. Int J Environ Res Public Health. 2020. PMID: 32842655 Free PMC article. Review.
-
A Practical Approach to Filtering Facepiece Respirator Decontamination and Reuse: Ultraviolet Germicidal Irradiation.Curr Treat Options Infect Dis. 2021;13(2):35-46. doi: 10.1007/s40506-021-00247-8. Epub 2021 Apr 6. Curr Treat Options Infect Dis. 2021. PMID: 33841050 Free PMC article. Review.
Cited by
-
Efficacy of Dry Heat Treatment against Clostridioides difficile Spores and Mycobacterium tuberculosis on Filtering Facepiece Respirators.Pathogens. 2022 Aug 2;11(8):871. doi: 10.3390/pathogens11080871. Pathogens. 2022. PMID: 36014991 Free PMC article.
-
End-user acceptability of personal protective equipment disinfection for potential reuse: a survey of health-care workers in Aotearoa New Zealand.Lancet Planet Health. 2023 Feb;7(2):e118-e127. doi: 10.1016/S2542-5196(22)00333-3. Lancet Planet Health. 2023. PMID: 36754468 Free PMC article.
-
Prominent Anti-UVC Activity of Lignin Degradation Products.In Vivo. 2022 Nov-Dec;36(6):2689-2699. doi: 10.21873/invivo.13004. In Vivo. 2022. PMID: 36309360 Free PMC article.
-
Comparison of UVC Sensitivity and Dectin-2 Expression Between Malignant and Non-malignant Cells.In Vivo. 2022 Sep-Oct;36(5):2116-2125. doi: 10.21873/invivo.12937. In Vivo. 2022. PMID: 36099100 Free PMC article.
References
-
- Basu I., Nagappan R., Fox-Lewis S., Muttaiyah S., McAuliffe G. Evaluation of extraction and amplification assays for the detection of SARS-CoV-2 at Auckland Hospital laboratory during the COVID-19 outbreak in New Zealand. J. Virol. Methods. 2021;289:114042. doi: 10.1016/j.jviromet.2020.114042. - DOI - PMC - PubMed
-
- Douglas J., Geoghegan J.L., Hadfield J., Bouckaert R., Storey M., Ren X., de Ligt J., French N., Welch D. Real-Time Genomics for Tracking Severe Acute Respiratory Syndrome Coronavirus 2 Border Incursions after Virus Elimination, New Zealand. Emerg. Infect. Dis. 2021;27:2361–2368. doi: 10.3201/eid2709.211097. - DOI - PMC - PubMed
-
- Jefferies S., French N., Gilkison C., Graham G., Hope V., Marshall J., McElnay C., McNeill A., Muellner P., Paine S., et al. COVID-19 in New Zealand and the impact of the national response: A descriptive epidemiological study. Lancet Public Health. 2020;5:e612–e623. doi: 10.1016/S2468-2667(20)30225-5. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

