Polatuzumab Vedotin in Previously Untreated Diffuse Large B-Cell Lymphoma
- PMID: 34904799
- PMCID: PMC11702892
- DOI: 10.1056/NEJMoa2115304
Polatuzumab Vedotin in Previously Untreated Diffuse Large B-Cell Lymphoma
Abstract
Background: Diffuse large B-cell lymphoma (DLBCL) is typically treated with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP). However, only 60% of patients are cured with R-CHOP. Polatuzumab vedotin is an antibody-drug conjugate targeting CD79b, which is ubiquitously expressed on the surface of malignant B cells.
Methods: We conducted a double-blind, placebo-controlled, international phase 3 trial to evaluate a modified regimen of R-CHOP (pola-R-CHP), in which vincristine was replaced with polatuzumab vedotin, as compared with standard R-CHOP, in patients with previously untreated intermediate-risk or high-risk DLBCL. Patients 18 to 80 years of age were randomly assigned in a 1:1 ratio to receive six cycles of either pola-R-CHP or R-CHOP, plus two cycles of rituximab alone. The primary end point was investigator-assessed progression-free survival. Secondary end points included overall survival and safety.
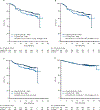
Results: Overall, 879 patients underwent randomization: 440 were assigned to the pola-R-CHP group and 439 to the R-CHOP group. After a median follow-up of 28.2 months, the percentage of patients surviving without progression was significantly higher in the pola-R-CHP group than in the R-CHOP group (76.7% [95% confidence interval (CI), 72.7 to 80.8] vs. 70.2% [95% CI, 65.8 to 74.6] at 2 years; stratified hazard ratio for progression, relapse, or death, 0.73 by Cox regression; 95% CI, 0.57 to 0.95; P = 0.02). Overall survival at 2 years did not differ significantly between the groups (88.7% [95% CI, 85.7 to 91.6] in the pola-R-CHP group and 88.6% [95% CI, 85.6 to 91.6] in the R-CHOP group; hazard ratio for death, 0.94; 95% CI, 0.65 to 1.37; P = 0.75). The safety profile was similar in the two groups.
Conclusions: Among patients with previously untreated intermediate-risk or high-risk DLBCL, the risk of disease progression, relapse, or death was lower among those who received pola-R-CHP than among those who received R-CHOP. (Funded by F. Hoffmann-La Roche/Genentech; POLARIX ClinicalTrials.gov number, NCT03274492.).
Copyright © 2021 Massachusetts Medical Society.
Figures
Comment in
-
All roads lead to targeted diffuse large B-cell lymphoma approaches.Cancer Cell. 2022 Feb 14;40(2):131-133. doi: 10.1016/j.ccell.2022.01.013. Cancer Cell. 2022. PMID: 35167823
Similar articles
-
Efficacy and Safety of Polatuzumab Vedotin Plus Rituximab, Cyclophosphamide, Doxorubicin and Prednisone for Previously Untreated Diffuse Large B-Cell Lymphoma: A Real-World, Multi-Center, Retrospective Cohort Study.Hematol Oncol. 2025 Jan;43(1):e70017. doi: 10.1002/hon.70017. Hematol Oncol. 2025. PMID: 39641321
-
FDA Approval Summary: Polatuzumab Vedotin in the First-Line Treatment of Select Large B-Cell Lymphomas.Clin Cancer Res. 2024 Dec 16;30(24):5521-5526. doi: 10.1158/1078-0432.CCR-24-1729. Clin Cancer Res. 2024. PMID: 39404868
-
Is first-line treatment with polatuzumab vedotin-rituximab-cyclophosphamide, doxorubicin and prednisone (pola-R-CHP) for previously untreated diffuse large B-cell lymphoma cost-effective in China? A cost-effectiveness analysis using a Markov model.BMJ Open. 2025 Jan 20;15(1):e086251. doi: 10.1136/bmjopen-2024-086251. BMJ Open. 2025. PMID: 39832983 Free PMC article.
-
Daratumumab and antineoplastic therapy versus antineoplastic therapy only for adults with newly diagnosed multiple myeloma ineligible for transplant.Cochrane Database Syst Rev. 2024 May 2;5(5):CD013595. doi: 10.1002/14651858.CD013595.pub2. Cochrane Database Syst Rev. 2024. PMID: 38695605 Review.
-
Impact of residual disease as a prognostic factor for survival in women with advanced epithelial ovarian cancer after primary surgery.Cochrane Database Syst Rev. 2022 Sep 26;9(9):CD015048. doi: 10.1002/14651858.CD015048.pub2. Cochrane Database Syst Rev. 2022. PMID: 36161421 Free PMC article. Review.
Cited by
-
Introduction of Carbonyl Groups into Antibodies.Molecules. 2023 Dec 1;28(23):7890. doi: 10.3390/molecules28237890. Molecules. 2023. PMID: 38067618 Free PMC article. Review.
-
Targeted and cellular therapies in lymphoma: Mechanisms of escape and innovative strategies.Front Oncol. 2022 Sep 12;12:948513. doi: 10.3389/fonc.2022.948513. eCollection 2022. Front Oncol. 2022. PMID: 36172151 Free PMC article. Review.
-
CNS prophylaxis in aggressive B-cell lymphoma.Hematology Am Soc Hematol Educ Program. 2022 Dec 9;2022(1):138-145. doi: 10.1182/hematology.2022000331. Hematology Am Soc Hematol Educ Program. 2022. PMID: 36485105 Free PMC article. Review.
-
Superiority of polatuzumab vedotin over other novel agents in previously untreated ABC-type diffuse large B-cell lymphoma: a network meta-analysis of 20 RCTs.Ann Hematol. 2023 May;102(5):1011-1017. doi: 10.1007/s00277-023-05161-1. Epub 2023 Mar 22. Ann Hematol. 2023. PMID: 36947214 Review.
-
The progress of novel strategies on immune-based therapy in relapsed or refractory diffuse large B-cell lymphoma.Exp Hematol Oncol. 2023 Aug 14;12(1):72. doi: 10.1186/s40164-023-00432-z. Exp Hematol Oncol. 2023. PMID: 37580826 Free PMC article. Review.
References
-
- Teras LR, DeSantis CE, Cerhan JR, Morton LM, Jemal A, Flowers CR. 2016 US lymphoid malignancy statistics by World Health Organization subtypes. CA Cancer J Clin 2016;66:443–59. - PubMed
-
- Coiffier B, Lepage E, Briere J, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med 2002;346:235–42. - PubMed
-
- Pfreundschuh M, Kuhnt E, Trümper L, et al. CHOP-like chemotherapy with or without rituximab in young patients with good-prognosis diffuse large-B-cell lymphoma: 6-year results of an open-label randomised study of the MabThera International Trial (MInT) Group. The Lancet Oncology 2011;12:1013–22. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
