Root-Associated Bacteria Community Characteristics of Antarctic Plants: Deschampsia antarctica and Colobanthus quitensis-a Comparison
- PMID: 34661728
- PMCID: PMC9622554
- DOI: 10.1007/s00248-021-01891-9
Root-Associated Bacteria Community Characteristics of Antarctic Plants: Deschampsia antarctica and Colobanthus quitensis-a Comparison
Abstract
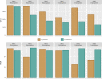
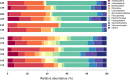
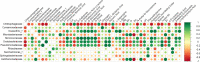
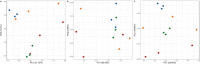
Colobanthus quitensis (Kunth) Bartl. and Deschampsia antarctica Desv. are the only Magnoliophyta to naturally colonize the Antarctic region. The reason for their sole presence in Antarctica is still debated as there is no definitive consensus on how only two unrelated flowering plants managed to establish breeding populations in this part of the world. In this study, we have explored and compared the rhizosphere and root-endosphere dwelling microbial community of C. quitensis and D. antarctica specimens sampled in maritime Antarctica from sites displaying contrasting edaphic characteristics. Bacterial phylogenetic diversity (high-throughput 16S rRNA gene fragment targeted sequencing) and microbial metabolic activity (Biolog EcoPlates) with a geochemical soil background were assessed. Gathered data showed that the microbiome of C. quitensis root system was mostly site-dependent, displaying different characteristics in each of the examined locations. This plant tolerated an active bacterial community only in severe conditions (salt stress and nutrient deprivation), while in other more favorable circumstances, it restricted microbial activity, with a possibility of microbivory-based nutrient acquisition. The microbial communities of D. antarctica showed a high degree of similarity between samples within a particular rhizocompartment. The grass' endosphere was significantly enriched in plant beneficial taxa of the family Rhizobiaceae, which displayed obligatory endophyte characteristics, suggesting that at least part of this community is transmitted vertically. Ultimately, the ecological success of C. quitensis and D. antarctica in Antarctica might be largely attributed to their associations and management of root-associated microbiota.
Keywords: Antarctic bacteria; Endosphere; Functional symbiosis; Microbial diversity; Rhizosphere.
© 2021. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Bacterial diversity in rhizosphere soil from Antarctic vascular plants of Admiralty Bay, maritime Antarctica.ISME J. 2010 Aug;4(8):989-1001. doi: 10.1038/ismej.2010.35. Epub 2010 Apr 1. ISME J. 2010. PMID: 20357834
-
Are Deschampsia antarctica Desv. and Colobanthus quitensis (Kunth) Bartl. migratory relicts?Tsitol Genet. 2007 Jul-Aug;41(4):36-40. Tsitol Genet. 2007. PMID: 18030724
-
Antarctic Hairgrass Rhizosphere Microbiomes: Microscale Effects Shape Diversity, Structure, and Function.Microbes Environ. 2022;37(2):ME21069. doi: 10.1264/jsme2.ME21069. Microbes Environ. 2022. PMID: 35705309 Free PMC article.
-
Origins of native vascular plants of Antarctica: comments from a historical phytogeography viewpoint.Tsitol Genet. 2007 Sep-Oct;41(5):54-63. Tsitol Genet. 2007. PMID: 18268968 Review.
-
Ecophysiology of Antarctic Vascular Plants: An Update on the Extreme Environment Resistance Mechanisms and Their Importance in Facing Climate Change.Plants (Basel). 2024 Feb 3;13(3):449. doi: 10.3390/plants13030449. Plants (Basel). 2024. PMID: 38337983 Free PMC article. Review.
Cited by
-
Endophytic Bacterial Biofilm-Formers Associated with Antarctic Vascular Plants.Microorganisms. 2024 Sep 25;12(10):1938. doi: 10.3390/microorganisms12101938. Microorganisms. 2024. PMID: 39458248 Free PMC article.
-
Soil warming increases the number of growing bacterial taxa but not their growth rates.Sci Adv. 2024 Feb 23;10(8):eadk6295. doi: 10.1126/sciadv.adk6295. Epub 2024 Feb 23. Sci Adv. 2024. PMID: 38394199 Free PMC article.
-
Polar-Region Soils as Novel Reservoir of Lactic Acid Bacteria from the Genus Carnobacterium.Int J Mol Sci. 2024 Aug 30;25(17):9444. doi: 10.3390/ijms25179444. Int J Mol Sci. 2024. PMID: 39273391 Free PMC article.
-
Simulated global warming affects endophytic bacterial and fungal communities of Antarctic pearlwort leaves and some bacterial isolates support plant growth at low temperatures.Sci Rep. 2022 Nov 6;12(1):18839. doi: 10.1038/s41598-022-23582-2. Sci Rep. 2022. PMID: 36336707 Free PMC article.
-
Source and acquisition of rhizosphere microbes in Antarctic vascular plants.Front Microbiol. 2022 Sep 8;13:916210. doi: 10.3389/fmicb.2022.916210. eCollection 2022. Front Microbiol. 2022. PMID: 36160194 Free PMC article.
References
-
- Pearce DA (2012) Extremophiles in Antarctica: life at low temperatures. In: Stan-Lotter H, Fendrihan S (eds) Adaption of microbial life to environmental extremes (pp. 87–118). Springer, Vienna. pp. 87–118
-
- Biersma EM, Torres-Díaz C, Molina-Montenegro MA, Newsham KK, Vidal MA, Collado GA, Acuña-Rodríguez IS, Ballesteros GI, Figueroa ChC, Goodall-Copestake WP, Leppe MA, Cuba-Díaz M, Valladares MA, Pertierra LR, Convey P. Multiple late-Pleistocene colonisation events of the Antarctic pearlwort Colobanthus quitensis (Caryophyllaceae) reveal the recent arrival of native Antarctic vascular flora. J Bioge. 2020;47(8):1663–1673. doi: 10.1111/jbi.13843. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

