Phase 2 Randomized Study of Oral Ibrexafungerp Versus Fluconazole in Vulvovaginal Candidiasis
- PMID: 34555149
- PMCID: PMC9258939
- DOI: 10.1093/cid/ciab841
Phase 2 Randomized Study of Oral Ibrexafungerp Versus Fluconazole in Vulvovaginal Candidiasis
Abstract
Background: Vulvovaginal candidiasis affects approximately 75% of women in their lifetime. Approved treatment options are limited to oral or topical azoles. Ibrexafungerp, a novel, first-in-class oral triterpenoid glucan synthase inhibitor, has demonstrated broad fungicidal Candida activity and a favorable tolerability profile. The primary objective of this dose-finding study was to identify the optimal dose of oral ibrexafungerp in patients with acute vulvovaginal candidiasis.
Methods: Patients with vulvovaginal signs and symptoms score ≥7 were randomized equally to 6 treatments groups: 5 treatment doses of oral ibrexafungerp or oral fluconazole 150 mg. The primary endpoint was the percentage of patients with a clinical cure (complete resolution of vulvovaginal signs and symptoms) at the test-of-cure visit (day 10).
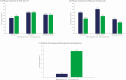
Results: Overall, 186 patients were randomized into the 6 treatment groups. Results, using the modified intent-to-treat population (baseline positive culture), are reported for ibrexafungerp 300 mg twice daily (BID) for 1 day (n = 27), which was the dose selected for phase 3 studies, and fluconazole 150 mg for 1 day (n = 24). At day 10, the clinical cure rates for ibrexafungerp and fluconazole were 51.9% and 58.3%, respectively; at day 25, patients with no signs or symptoms were 70.4% and 50.0%, respectively. During the study ibrexafungerp patients required less antifungal rescue medications compared with fluconazole (3.7% vs 29.2%, respectively). Ibrexafungerp was well tolerated, with the most common treatment-related adverse events being mild gastrointestinal events.
Conclusions: Ibrexafungerp is a well-tolerated novel antifungal with comparable efficacy to fluconazole in the treatment of acute vulvovaginal candidiasis.
Clinical trials registration: NCT03253094.
Keywords: Candida albicans; SCY-078; fluconazole; ibrexafungerp; vulvovaginal candidiasis.
© The Author(s) 2021. Published by Oxford University Press for the Infectious Diseases Society of America.
Figures
Similar articles
-
Efficacy and safety of oral ibrexafungerp in Chinese patients with vulvovaginal candidiasis: a phase III, randomized, double-blind study.Infection. 2024 Oct;52(5):1787-1797. doi: 10.1007/s15010-024-02233-w. Epub 2024 Apr 3. Infection. 2024. PMID: 38568411 Free PMC article. Clinical Trial.
-
Ibrexafungerp Versus Placebo for Vulvovaginal Candidiasis Treatment: A Phase 3, Randomized, Controlled Superiority Trial (VANISH 303).Clin Infect Dis. 2022 Jun 10;74(11):1979-1985. doi: 10.1093/cid/ciab750. Clin Infect Dis. 2022. PMID: 34467969 Free PMC article. Clinical Trial.
-
Oral Ibrexafungerp for Vulvovaginal Candidiasis Treatment: An Analysis of VANISH 303 and VANISH 306.J Womens Health (Larchmt). 2023 Feb;32(2):178-186. doi: 10.1089/jwh.2022.0132. Epub 2022 Oct 17. J Womens Health (Larchmt). 2023. PMID: 36255448 Free PMC article. Clinical Trial.
-
Ibrexafungerp for the Treatment of Vulvovaginal Candidiasis: Design, Development and Place in Therapy.Drug Des Devel Ther. 2023 Feb 7;17:363-367. doi: 10.2147/DDDT.S339349. eCollection 2023. Drug Des Devel Ther. 2023. PMID: 36785761 Free PMC article. Review.
-
Ibrexafungerp in the Treatment of Vulvovaginal Candidiasis.Ann Pharmacother. 2023 Jan;57(1):99-106. doi: 10.1177/10600280221091301. Epub 2022 May 2. Ann Pharmacother. 2023. PMID: 35502451 Review.
Cited by
-
A review of the fernane-type triterpenoids as anti-fungal drugs.Front Pharmacol. 2024 Aug 21;15:1447450. doi: 10.3389/fphar.2024.1447450. eCollection 2024. Front Pharmacol. 2024. PMID: 39234110 Free PMC article. Review.
-
Antifungal chemotherapies and immunotherapies for the future.Parasite Immunol. 2023 Feb;45(2):e12960. doi: 10.1111/pim.12960. Epub 2022 Dec 18. Parasite Immunol. 2023. PMID: 36403106 Free PMC article. Review.
-
Ibrexafungerp to treat acute vulvovaginal candidiasis.Nat Rev Urol. 2021 Nov;18(11):638. doi: 10.1038/s41585-021-00522-9. Nat Rev Urol. 2021. PMID: 34548655 No abstract available.
-
Novel antifungals and treatment approaches to tackle resistance and improve outcomes of invasive fungal disease.Clin Microbiol Rev. 2024 Jun 13;37(2):e0007423. doi: 10.1128/cmr.00074-23. Epub 2024 Apr 11. Clin Microbiol Rev. 2024. PMID: 38602408 Review.
-
Efficacy and safety of oral ibrexafungerp in Chinese patients with vulvovaginal candidiasis: a phase III, randomized, double-blind study.Infection. 2024 Oct;52(5):1787-1797. doi: 10.1007/s15010-024-02233-w. Epub 2024 Apr 3. Infection. 2024. PMID: 38568411 Free PMC article. Clinical Trial.
References
-
- Mendling W, Brasch J, Cornely OA, et al. . Guideline: vulvovaginal candidosis (AWMF 015/072), S2k (excluding chronic mucocutaneous candidosis). Mycoses 2015; 58:1–15. - PubMed
-
- Jeanmonod R, Jeanmonod D.. Vaginal candidiasis. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing, 2020. Available at: https://www.ncbi.nlm.nih.gov/books/NBK459317/. Updated 21 November 2020. Accessed 9 December 2020.
-
- Committee on Practice Bulletins—Gynecology. Vaginitis in nonpregnant patients: ACOG practice bulletin, Number 215. Obstet Gynecol 2020; 135:e1–17. - PubMed

