Evaluation of reporting quality of randomized controlled trials in patients with COVID-19 using the CONSORT statement
- PMID: 34555033
- PMCID: PMC8460279
- DOI: 10.1371/journal.pone.0257093
Evaluation of reporting quality of randomized controlled trials in patients with COVID-19 using the CONSORT statement
Abstract
Objective: To evaluate the reporting quality of randomized controlled trials (RCTs) regarding patients with COVID-19 and analyse the influence factors.
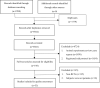
Methods: PubMed, Embase, Web of Science and the Cochrane Library databases were searched to collect RCTs regarding patients with COVID-19. The retrieval time was from the inception to December 1, 2020. The CONSORT 2010 statement was used to evaluate the overall reporting quality of these RCTs.
Results: 53 RCTs were included. The study showed that the average reporting rate for 37 items in CONSORT checklist was 53.85% with mean overall adherence score of 13.02±3.546 (ranged: 7 to 22). The multivariate linear regression analysis showed the overall adherence score to the CONSORT guideline was associated with journal impact factor (P = 0.006), and endorsement of CONSORT statement (P = 0.014).
Conclusion: Although many RCTs of COVID-19 have been published in different journals, the overall reporting quality of these articles was suboptimal, it can not provide valid evidence for clinical decision-making and systematic reviews. Therefore, more journals should endorse the CONSORT statement, authors should strictly follow the relevant provisions of the CONSORT guideline when reporting articles. Future RCTs should particularly focus on improvement of detailed reporting in allocation concealment, blinding and estimation of sample size.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
Consolidated standards of reporting trials (CONSORT) and the completeness of reporting of randomised controlled trials (RCTs) published in medical journals.Cochrane Database Syst Rev. 2012 Nov 14;11(11):MR000030. doi: 10.1002/14651858.MR000030.pub2. Cochrane Database Syst Rev. 2012. PMID: 23152285 Free PMC article. Review.
-
Assessment of the quality of reporting of randomised controlled trials in otorhinolaryngologic literature - adherence to the CONSORT statement.PLoS One. 2015 Mar 20;10(3):e0122328. doi: 10.1371/journal.pone.0122328. eCollection 2015. PLoS One. 2015. PMID: 25793517 Free PMC article.
-
Impact of the CONSORT Statement endorsement in the completeness of reporting of randomized clinical trials in restorative dentistry.J Dent. 2017 Mar;58:54-59. doi: 10.1016/j.jdent.2017.01.009. Epub 2017 Jan 31. J Dent. 2017. PMID: 28159507
-
Evaluation of reporting quality of abstracts of randomized controlled trials regarding patients with COVID-19 using the CONSORT statement for abstracts.Int J Infect Dis. 2022 Jan 7;116:122-129. doi: 10.1016/j.ijid.2022.01.002. Online ahead of print. Int J Infect Dis. 2022. PMID: 34999245 Free PMC article.
-
Does use of the CONSORT Statement impact the completeness of reporting of randomised controlled trials published in medical journals? A Cochrane review.Syst Rev. 2012 Nov 29;1:60. doi: 10.1186/2046-4053-1-60. Syst Rev. 2012. PMID: 23194585 Free PMC article. Review.
Cited by
-
Improved outcomes with perioperative dietitian-led interventions in patients undergoing total joint arthroplasty: A systematic review.J Orthop. 2024 Apr 29;56:12-17. doi: 10.1016/j.jor.2024.04.021. eCollection 2024 Oct. J Orthop. 2024. PMID: 38737733 Review.
-
Debunking the myth of using "quiet" in clinical departments: an integrative overview of available literature.Ann Med Surg (Lond). 2022 Oct 6;82:104792. doi: 10.1016/j.amsu.2022.104792. eCollection 2022 Oct. Ann Med Surg (Lond). 2022. PMID: 36268415 Free PMC article. No abstract available.
-
Poor reporting quality of randomized controlled trials comparing treatments of COVID-19-A retrospective cross-sectional study on the first year of publications.PLoS One. 2023 Oct 16;18(10):e0292860. doi: 10.1371/journal.pone.0292860. eCollection 2023. PLoS One. 2023. PMID: 37844082 Free PMC article.
-
Evaluating the completeness of the reporting of abstracts since the publication of the CONSORT extension for abstracts: an evaluation of randomized controlled trial in ten nursing journals.Trials. 2023 Jun 22;24(1):423. doi: 10.1186/s13063-023-07419-5. Trials. 2023. PMID: 37349754 Free PMC article.
-
Transparency and reporting characteristics of COVID-19 randomized controlled trials.BMC Med. 2022 Sep 26;20(1):363. doi: 10.1186/s12916-022-02567-y. BMC Med. 2022. PMID: 36154932 Free PMC article.
References
-
- Hajibandeh S, Hajibandeh S, Antoniou GA, Green PA, Maden M, Torella F. Reporting and Methodological Quality of Randomised Controlled Trials in Vascular and Endovascular Surgery. European journal of vascular and endovascular surgery: the official journal of the European Society for Vascular Surgery. 2015;50(5):664–70. doi: 10.1016/j.ejvs.2015.06.114 - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical