Exposure-Response Analyses for Therapeutic Dose Selection of Belantamab Mafodotin in Patients With Relapsed/Refractory Multiple Myeloma
- PMID: 34468979
- PMCID: PMC9293327
- DOI: 10.1002/cpt.2409
Exposure-Response Analyses for Therapeutic Dose Selection of Belantamab Mafodotin in Patients With Relapsed/Refractory Multiple Myeloma
Abstract
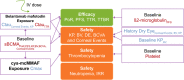
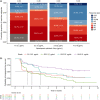
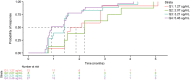
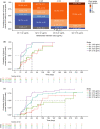
Belantamab mafodotin is an antibody-drug conjugate comprising a humanized anti-B-cell maturation antigen (BCMA) monoclonal antibody conjugated to monomethyl auristatin F (MMAF) via a protease-resistant maleimidocaproyl linker. Single-agent belantamab mafodotin showed clinically meaningful activity and manageable safety in patients with heavily pretreated relapsed/refractory multiple myeloma (RRMM) in the phase I DREAMM-1 and phase II DREAMM-2 studies and is approved by the US Food and Drug Administration and European Medicines Agency for RRMM treatment. To support monotherapy dose selection, the relationship between Cycle 1 exposure (derived using a population pharmacokinetic model) and clinical response (for multiple efficacy and safety end points) was explored. In DREAMM-2, efficacy end points (probability of response (PoR) and progression-free survival (PFS)) were associated with exposure in univariate evaluation; however, once disease burden factors were included in the model (e.g., baseline soluble BCMA, ß2 -microglobulin), exposure was no longer significant. Patients with higher disease burden had lower exposure. In DREAMM-1, belantamab mafodotin exposure was the only variable to correlate with PoR and PFS. Probability of corneal events (keratopathy), but not dry eye or blurred vision, was strongly associated with belantamab mafodotin exposure (DREAMM-2). Higher cys-mcMMAF maximum plasma drug concentration (Cmax ) and lower baseline platelet count were associated with increased probability of thrombocytopenia (DREAMM-1 and DREAMM -2). In general, safety end points were more strongly associated with belantamab mafodotin exposure than efficacy end points, particularly after disease factors and patient characteristics were taken into account. Overall, these findings supported the monotherapy dose recommendation of belantamab mafodotin as 2.5 mg/kg every 3 weeks in patients with RRMM who have received four or more prior therapies.
© 2021 Glaxo Group Limited. Clinical Pharmacology & Therapeutics published by Wiley Periodicals LLC on behalf of American Society for Clinical Pharmacology and Therapeutics.
Conflict of interest statement
G.F.‐B., C.R., J.C., H.S., S.V., and J.O. are employees of GlaxoSmithKline and hold ownership interests. R.C.J. is an employee of GlaxoSmithKline and holds ownership interests in GlaxoSmithKline and Novartis.
Figures
Similar articles
-
EMA Review of Belantamab Mafodotin (Blenrep) for the Treatment of Adult Patients with Relapsed/Refractory Multiple Myeloma.Oncologist. 2021 Jan;26(1):70-76. doi: 10.1002/onco.13592. Epub 2020 Nov 23. Oncologist. 2021. PMID: 33179377 Free PMC article. Clinical Trial.
-
Belantamab mafodotin concentration-QTc relationships in patients with relapsed or refractory multiple myeloma from the DREAMM-1 and -2 studies.Br J Clin Pharmacol. 2024 Oct;90(10):2571-2581. doi: 10.1111/bcp.16133. Epub 2024 Jun 23. Br J Clin Pharmacol. 2024. PMID: 38924122 Clinical Trial.
-
Population pharmacokinetics of belantamab mafodotin, a BCMA-targeting agent in patients with relapsed/refractory multiple myeloma.CPT Pharmacometrics Syst Pharmacol. 2021 Aug;10(8):851-863. doi: 10.1002/psp4.12660. Epub 2021 Jul 29. CPT Pharmacometrics Syst Pharmacol. 2021. PMID: 34076364 Free PMC article.
-
Belantamab Mafodotin: First Approval.Drugs. 2020 Oct;80(15):1607-1613. doi: 10.1007/s40265-020-01404-x. Drugs. 2020. PMID: 32936437 Review.
-
Belantamab Mafodotin for the Treatment of Multiple Myeloma: An Overview of the Clinical Efficacy and Safety.Drug Des Devel Ther. 2021 Jun 2;15:2401-2415. doi: 10.2147/DDDT.S267404. eCollection 2021. Drug Des Devel Ther. 2021. PMID: 34103900 Free PMC article. Review.
Cited by
-
Longitudinal efficacy and safety modeling and simulation framework to aid dose selection of belantamab mafodotin for patients with multiple myeloma.CPT Pharmacometrics Syst Pharmacol. 2023 Oct;12(10):1411-1424. doi: 10.1002/psp4.13016. Epub 2023 Aug 2. CPT Pharmacometrics Syst Pharmacol. 2023. PMID: 37465991 Free PMC article.
-
Drug conjugates for the treatment of lung cancer: from drug discovery to clinical practice.Exp Hematol Oncol. 2024 Mar 1;13(1):26. doi: 10.1186/s40164-024-00493-8. Exp Hematol Oncol. 2024. PMID: 38429828 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

