Micro- and Mycobiota Dysbiosis in Pancreatic Ductal Adenocarcinoma Development
- PMID: 34298645
- PMCID: PMC8303110
- DOI: 10.3390/cancers13143431
Micro- and Mycobiota Dysbiosis in Pancreatic Ductal Adenocarcinoma Development
Abstract
Background: Dysbiosis of the intestinal flora has emerged as an oncogenic contributor in different malignancies. Recent findings suggest a crucial tumor-promoting role of micro- and mycobiome alterations also in the development of pancreatic ductal adenocarcinoma (PDAC).
Methods: To summarize the current knowledge about this topic, a systematic literature search of articles published until October 2020 was performed in MEDLINE (PubMed).
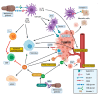
Results: An increasing number of publications describe associations between bacterial and fungal species and PDAC development. Despite the high inter-individual variability of the commensal flora, some studies identify specific microbial signatures in PDAC patients, including oral commensals like Porphyromonas gingivalis and Fusobacterium nucleatum or Gram-negative bacteria like Proteobacteria. The role of Helicobacter spp. remains unclear. Recent isolation of Malassezia globosa from PDAC tissue suggest also the mycobiota as a crucial player of tumorigenesis. Based on described molecular mechanisms and interactions between the pancreatic tissue and the immune system this review proposes a model of how the micro- and the mycobial dysbiosis could contribute to tumorigenesis in PDAC.
Conclusions: The presence of micro- and mycobial dysbiosis in pancreatic tumor tissue opens a fascinating perspective on PDAC oncogenesis. Further studies will pave the way for novel tumor markers and treatment strategies.
Keywords: Malassezia; Proteobacteria; immunosuppression; inflammation; microbiome; mycobiome; pancreatic cancer; tumor initiation; tumor progression.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Complement and Fungal Dysbiosis as Prognostic Markers and Potential Targets in PDAC Treatment.Curr Oncol. 2022 Dec 14;29(12):9833-9854. doi: 10.3390/curroncol29120773. Curr Oncol. 2022. PMID: 36547187 Free PMC article. Review.
-
Fungi, host immune response, and tumorigenesis.Am J Physiol Gastrointest Liver Physiol. 2021 Aug 1;321(2):G213-G222. doi: 10.1152/ajpgi.00025.2021. Epub 2021 Jul 7. Am J Physiol Gastrointest Liver Physiol. 2021. PMID: 34231392 Free PMC article. Review.
-
The Microbiome as a Potential Target for Therapeutic Manipulation in Pancreatic Cancer.Cancers (Basel). 2021 Jul 27;13(15):3779. doi: 10.3390/cancers13153779. Cancers (Basel). 2021. PMID: 34359684 Free PMC article. Review.
-
The Emerging Role of Microbiota and Microbiome in Pancreatic Ductal Adenocarcinoma.Biomedicines. 2020 Dec 3;8(12):565. doi: 10.3390/biomedicines8120565. Biomedicines. 2020. PMID: 33287196 Free PMC article. Review.
-
Microbial Associations with Pancreatic Cancer: A New Frontier in Biomarkers.Cancers (Basel). 2021 Jul 27;13(15):3784. doi: 10.3390/cancers13153784. Cancers (Basel). 2021. PMID: 34359685 Free PMC article. Review.
Cited by
-
IPIAD- an augmentation regimen added to standard treatment of pancreatic ductal adenocarcinoma using already-marketed repurposed drugs irbesartan, pyrimethamine, itraconazole, azithromycin, and dapsone.Oncoscience. 2024 Feb 7;11:15-31. doi: 10.18632/oncoscience.594. eCollection 2024. Oncoscience. 2024. PMID: 38524376 Free PMC article.
-
The inflammatory response of human pancreatic cancer samples compared to normal controls.PLoS One. 2023 Nov 1;18(11):e0284232. doi: 10.1371/journal.pone.0284232. eCollection 2023. PLoS One. 2023. PMID: 37910468 Free PMC article.
-
Intratumoral microbiota: implications for cancer onset, progression, and therapy.Front Immunol. 2024 Jan 16;14:1301506. doi: 10.3389/fimmu.2023.1301506. eCollection 2023. Front Immunol. 2024. PMID: 38292482 Free PMC article. Review.
-
Complement and Fungal Dysbiosis as Prognostic Markers and Potential Targets in PDAC Treatment.Curr Oncol. 2022 Dec 14;29(12):9833-9854. doi: 10.3390/curroncol29120773. Curr Oncol. 2022. PMID: 36547187 Free PMC article. Review.
-
The role of the symbiotic microecosystem in cancer: gut microbiota, metabolome, and host immunome.Front Immunol. 2023 Aug 24;14:1235827. doi: 10.3389/fimmu.2023.1235827. eCollection 2023. Front Immunol. 2023. PMID: 37691931 Free PMC article. Review.
References
-
- Neoptolemos J.P., Palmer D.H., Ghaneh P., Psarelli E.E., Valle J.W., Halloran C.M., Faluyi O., O’Reilly D.A., Cunningham D., Wadsley J., et al. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): A multicentre, open-label, randomised, phase 3 trial. Lancet. 2017;389:1011–1024. doi: 10.1016/S0140-6736(16)32409-6. - DOI - PubMed
-
- Uesaka K., Boku N., Fukutomi A., Okamura Y., Konishi M., Matsumoto I., Kaneoka Y., Shimizu Y., Nakamori S., Sakamoto H., et al. Adjuvant chemotherapy of S-1 versus gemcitabine for resected pancreatic cancer: A phase 3, open-label, randomised, non-inferiority trial (JASPAC 01) Lancet. 2016;388:248–257. doi: 10.1016/S0140-6736(16)30583-9. - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

