Long-term outcomes from the Phase II L-MIND study of tafasitamab (MOR208) plus lenalidomide in patients with relapsed or refractory diffuse large B-cell lymphoma
- PMID: 34196165
- PMCID: PMC8409029
- DOI: 10.3324/haematol.2020.275958
Long-term outcomes from the Phase II L-MIND study of tafasitamab (MOR208) plus lenalidomide in patients with relapsed or refractory diffuse large B-cell lymphoma
Abstract
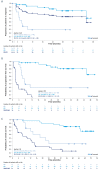
Tafasitamab (MOR208), an Fc-modified, humanized, anti-CD19 monoclonal antibody, combined with the immunomodulatory drug lenalidomide was clinically active with a good tolerability profile in the open-label, single-arm, phase II L-MIND study of patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL) ineligible for autologous stem-cell transplantation. To assess long-term outcomes, we report an updated analysis with ≥35 months' follow-up. Patients were aged >18 years, had received one to three prior systemic therapies (including ≥1 CD20-targeting regimen) and Eastern Cooperative Oncology Group performance status 0-2. Patients received 28-day cycles of tafasitamab (12 mg/kg intravenously), once weekly during cycles 1-3, then every 2 weeks during cycles 4-12. Lenalidomide (25 mg orally) was administered on days 1-21 of cycles 1-12. After cycle 12, progression-free patients received tafasitamab every 2 weeks until disease progression. The primary endpoint was best objective response rate. After ≥35 months' follow-up (data cut-off: October 30, 2020), the objective response rate was 57.5% (n=46/80), including a complete response in 40.0% of patients (n=32/80) and a partial response in 17.5% of patients (n=14/80). The median duration of response was 43.9 months (95% confidence interval [95% CI]: 26.1-not reached), the median overall survival was 33.5 months (95% CI: 18.3-not reached) and the median progression-free survival was 11.6 months (95% CI: 6.3-45.7). There were no unexpected toxicities. Subgroup analyses revealed consistent long-term efficacy results across most subgroups of patients. This extended follow-up of L-MIND confirms the long duration of response, meaningful overall survival, and well-defined safety profile of tafasitamab plus lenalidomide followed by tafasitamab monotherapy in patients with relapsed/refractory diffuse large B-cell lymphoma ineligible for autologous stem cell transplantation. ClinicalTrials.gov identifier: NCT02399085.
Figures

Similar articles
-
Tafasitamab plus lenalidomide in relapsed or refractory diffuse large B-cell lymphoma (L-MIND): a multicentre, prospective, single-arm, phase 2 study.Lancet Oncol. 2020 Jul;21(7):978-988. doi: 10.1016/S1470-2045(20)30225-4. Epub 2020 Jun 5. Lancet Oncol. 2020. PMID: 32511983 Clinical Trial.
-
Tafasitamab for patients with relapsed or refractory diffuse large B-cell lymphoma: final 5-year efficacy and safety findings in the phase II L-MIND study.Haematologica. 2024 Feb 1;109(2):553-566. doi: 10.3324/haematol.2023.283480. Haematologica. 2024. PMID: 37646664 Free PMC article. Clinical Trial.
-
Tafasitamab Plus Lenalidomide Versus 3 Rituximab-Based Treatments for Non-Transplant Eligible Relapsed/Refractory Diffuse Large B-Cell Lymphoma: A Matching-Adjusted Indirect Comparison.Adv Ther. 2022 Jun;39(6):2668-2687. doi: 10.1007/s12325-022-02094-5. Epub 2022 Apr 11. Adv Ther. 2022. PMID: 35403948 Free PMC article.
-
Tafasitamab for the treatment of relapsed or refractory diffuse large B-cell lymphoma.Expert Opin Biol Ther. 2021 Apr;21(4):455-463. doi: 10.1080/14712598.2021.1884677. Epub 2021 Feb 15. Expert Opin Biol Ther. 2021. PMID: 33554668 Free PMC article. Review.
-
Tafasitamab: First Approval.Drugs. 2020 Nov;80(16):1731-1737. doi: 10.1007/s40265-020-01405-w. Drugs. 2020. PMID: 32946059 Review.
Cited by
-
Targeted Treatment of Adults with Relapsed or Refractory Diffuse Large B-Cell Lymphoma (DLBCL): Tafasitamab in Context.Onco Targets Ther. 2023 Jul 20;16:617-629. doi: 10.2147/OTT.S372783. eCollection 2023. Onco Targets Ther. 2023. PMID: 37492075 Free PMC article. Review.
-
New treatment options in elderly patients with Diffuse Large B-cell Lymphoma.Front Oncol. 2023 Jul 3;13:1214026. doi: 10.3389/fonc.2023.1214026. eCollection 2023. Front Oncol. 2023. PMID: 37465115 Free PMC article. Review.
-
[Current researches and progresses on new antibody drugs in the treatment of relapsed/refractory diffuse large B-cell lymphoma].Zhonghua Xue Ye Xue Za Zhi. 2023 Mar 14;44(3):259-264. doi: 10.3760/cma.j.issn.0253-2727.2023.03.016. Zhonghua Xue Ye Xue Za Zhi. 2023. PMID: 37356992 Free PMC article. Chinese. No abstract available.
-
Relapsed/Refractory Diffuse Large B-Cell Lymphoma: A Look at the Approved and Emerging Therapies.J Pers Med. 2021 Dec 10;11(12):1345. doi: 10.3390/jpm11121345. J Pers Med. 2021. PMID: 34945817 Free PMC article. Review.
-
Current options and future perspectives in the treatment of patients with relapsed/refractory diffuse large B-cell lymphoma.Ther Adv Hematol. 2022 Jun 28;13:20406207221103321. doi: 10.1177/20406207221103321. eCollection 2022. Ther Adv Hematol. 2022. PMID: 35785244 Free PMC article. Review.
References
-
- World Health Organization. World Cancer Report 2020: Cancer Research for Cancer Prevention. Cancer Control. 2020;199.
-
- Sarkozy C, Sehn LH. New drugs for the management of relapsed or refractory diffuse large B-cell lymphoma. Ann Lymphoma. 2019;3:10.
-
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines: B-Cell Lymphomas V3.2021. Published 2021. https://www.nccn.org/professionals/physician_gls/pdf/b-cell.pdf. Accessed February 7, 2021.
-
- Tilly H, Gomes da Silva M, Vitolo U, et al. . Diffuse large B-cell lymphoma (DLBCL): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2015;26(Suppl 5):v116-v125. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources

