Unique variants in CLCN3, encoding an endosomal anion/proton exchanger, underlie a spectrum of neurodevelopmental disorders
- PMID: 34186028
- PMCID: PMC8387284
- DOI: 10.1016/j.ajhg.2021.06.003
Unique variants in CLCN3, encoding an endosomal anion/proton exchanger, underlie a spectrum of neurodevelopmental disorders
Abstract
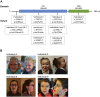
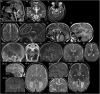
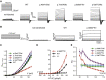
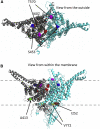
The genetic causes of global developmental delay (GDD) and intellectual disability (ID) are diverse and include variants in numerous ion channels and transporters. Loss-of-function variants in all five endosomal/lysosomal members of the CLC family of Cl- channels and Cl-/H+ exchangers lead to pathology in mice, humans, or both. We have identified nine variants in CLCN3, the gene encoding CIC-3, in 11 individuals with GDD/ID and neurodevelopmental disorders of varying severity. In addition to a homozygous frameshift variant in two siblings, we identified eight different heterozygous de novo missense variants. All have GDD/ID, mood or behavioral disorders, and dysmorphic features; 9/11 have structural brain abnormalities; and 6/11 have seizures. The homozygous variants are predicted to cause loss of ClC-3 function, resulting in severe neurological disease similar to the phenotype observed in Clcn3-/- mice. Their MRIs show possible neurodegeneration with thin corpora callosa and decreased white matter volumes. Individuals with heterozygous variants had a range of neurodevelopmental anomalies including agenesis of the corpus callosum, pons hypoplasia, and increased gyral folding. To characterize the altered function of the exchanger, electrophysiological analyses were performed in Xenopus oocytes and mammalian cells. Two variants, p.Ile607Thr and p.Thr570Ile, had increased currents at negative cytoplasmic voltages and loss of inhibition by luminal acidic pH. In contrast, two other variants showed no significant difference in the current properties. Overall, our work establishes a role for CLCN3 in human neurodevelopment and shows that both homozygous loss of ClC-3 and heterozygous variants can lead to GDD/ID and neuroanatomical abnormalities.
Keywords: CLCN; acidification; gain of function; hippocampus; intellectual disability; neurodevelopmental delay; pH sensitivity; voltage gated chloride channel.
Copyright © 2021 American Society of Human Genetics. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
P.B.A. is on the Scientific Advisory Board of Illumina, Inc. and GeneDx. The other authors declare no competing interests.
Figures


Similar articles
-
Uncoupling endosomal CLC chloride/proton exchange causes severe neurodegeneration.EMBO J. 2020 May 4;39(9):e103358. doi: 10.15252/embj.2019103358. Epub 2020 Mar 2. EMBO J. 2020. PMID: 32118314 Free PMC article.
-
Expanding the genetic and phenotypic relevance of CLCN4 variants in neurodevelopmental condition: 13 new patients.J Neurol. 2024 Aug;271(8):4933-4948. doi: 10.1007/s00415-024-12383-4. Epub 2024 May 17. J Neurol. 2024. PMID: 38758281
-
De novo CLCN3 variants affecting Gly327 cause severe neurodevelopmental syndrome with brain structural abnormalities.J Hum Genet. 2023 Apr;68(4):291-298. doi: 10.1038/s10038-022-01106-0. Epub 2022 Dec 20. J Hum Genet. 2023. PMID: 36536096
-
Neurodegeneration Upon Dysfunction of Endosomal/Lysosomal CLC Chloride Transporters.Front Cell Dev Biol. 2021 Feb 23;9:639231. doi: 10.3389/fcell.2021.639231. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33708769 Free PMC article. Review.
-
Cell biology and physiology of CLC chloride channels and transporters.Compr Physiol. 2012 Jul;2(3):1701-44. doi: 10.1002/cphy.c110038. Compr Physiol. 2012. PMID: 23723021 Review.
Cited by
-
Vesicular CLC chloride/proton exchangers in health and diseases.Front Pharmacol. 2023 Nov 7;14:1295068. doi: 10.3389/fphar.2023.1295068. eCollection 2023. Front Pharmacol. 2023. PMID: 38027030 Free PMC article. Review.
-
Structural basis of adenine nucleotides regulation and neurodegenerative pathology in ClC-3 exchanger.Nat Commun. 2024 Aug 6;15(1):6654. doi: 10.1038/s41467-024-50975-w. Nat Commun. 2024. PMID: 39107281 Free PMC article.
-
CryoEM structures of the human CLC-2 voltage gated chloride channel reveal a ball and chain gating mechanism.bioRxiv [Preprint]. 2023 Nov 29:2023.08.13.553136. doi: 10.1101/2023.08.13.553136. bioRxiv. 2023. Update in: Elife. 2024 Feb 12;12:RP90648. doi: 10.7554/eLife.90648. PMID: 37645939 Free PMC article. Updated. Preprint.
-
Identification of two novel autism genes, TRPC4 and SCFD2, in Qatar simplex families through exome sequencing.Front Psychiatry. 2023 Oct 31;14:1251884. doi: 10.3389/fpsyt.2023.1251884. eCollection 2023. Front Psychiatry. 2023. PMID: 38025430 Free PMC article.
-
ClC-3 regulates the excitability of nociceptive neurons and is involved in inflammatory processes within the spinal sensory pathway.Front Cell Neurosci. 2022 Aug 24;16:920075. doi: 10.3389/fncel.2022.920075. eCollection 2022. Front Cell Neurosci. 2022. PMID: 37124866 Free PMC article.
References
-
- Gilissen C., Hehir-Kwa J.Y., Thung D.T., van de Vorst M., van Bon B.W., Willemsen M.H., Kwint M., Janssen I.M., Hoischen A., Schenck A. Genome sequencing identifies major causes of severe intellectual disability. Nature. 2014;511:344–347. - PubMed
-
- Vissers L.E., Gilissen C., Veltman J.A. Genetic studies in intellectual disability and related disorders. Nat. Rev. Genet. 2016;17:9–18. - PubMed
-
- Jentsch T.J., Pusch M. CLC Chloride Channels and Transporters: Structure, Function, Physiology, and Disease. Physiol. Rev. 2018;98:1493–1590. - PubMed
-
- Lange P.F., Wartosch L., Jentsch T.J., Fuhrmann J.C. ClC-7 requires Ostm1 as a β-subunit to support bone resorption and lysosomal function. Nature. 2006;440:220–223. - PubMed
-
- Lloyd S.E., Pearce S.H., Fisher S.E., Steinmeyer K., Schwappach B., Scheinman S.J., Harding B., Bolino A., Devoto M., Goodyer P. A common molecular basis for three inherited kidney stone diseases. Nature. 1996;379:445–449. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

