Evaluation of 2 Ultraviolet-C Light Boxes for Decontamination of N95 Respirators
- PMID: 34136729
- PMCID: PMC8201793
- DOI: 10.20411/pai.v6i1.432
Evaluation of 2 Ultraviolet-C Light Boxes for Decontamination of N95 Respirators
Abstract
Background: Ultraviolet-C (UV-C) light devices are effective in reducing contamination on N95 filtering facepiece respirators. However, limited information is available on whether UV-C devices meet the Food and Drug Administration's (FDA) microbiological requirements for Emergency Use Authorization (EUA) for respirator bioburden reduction.
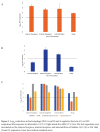
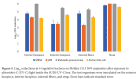
Methods: We tested the ability of 2 UV-C light boxes to achieve the 3-log10 microorganism reductions required for EUA for reuse by single users. Whole 3M 1860 or Moldex 1513 respirators were inoculated on the exterior facepiece, interior facepiece, and internal fibers with bacteriophage MS2 and/or 4 strains of bacteria and treated with UV-C cycles of 1 or 20 minutes. Colorimetric indicators were used to assess penetration of UV-C through the respirators.
Results: For 1 UV-C box, a 20-minute treatment achieved the required bioburden reduction for Moldex 1513 but not 3M 1860 respirators. For the second UV-C box, a 1-minute treatment achieved the required bioburden reduction in 4 bacterial strains for the Moldex 1513 respirator. Colorimetric indicators demonstrated penetration of UV-C through all layers of the Moldex 1513 respirator but not the 3M 1860 respirator.
Conclusions: Our findings demonstrate that UV-C box technologies can achieve bioburden reductions required by the FDA for EUA for single users but highlight the potential for variable efficacy for different types of respirators.
Keywords: N95 respirator; SARS-CoV-2; bacteriophage MS2; ultraviolet light.
Copyright © Pathogens and Immunity 2021.
Conflict of interest statement
C.J.D has received research grants from Clorox, Pfizer, and PDI. J.G.S. and I.C. have received patents for the Synchronous UV Decontamination System (SUDS). All other authors report no conflicts of interest relevant to this article.
Figures



Similar articles
-
Filtering Facepiece Respirator (N95 Respirator) Reprocessing: A Systematic Review.JAMA. 2021 Apr 6;325(13):1296-1317. doi: 10.1001/jama.2021.2531. JAMA. 2021. PMID: 33656543
-
The effect of ultraviolet C radiation against different N95 respirators inoculated with SARS-CoV-2.Int J Infect Dis. 2020 Nov;100:224-229. doi: 10.1016/j.ijid.2020.08.077. Epub 2020 Sep 3. Int J Infect Dis. 2020. PMID: 32891736 Free PMC article.
-
Principles and practice for SARS-CoV-2 decontamination of N95 masks with UV-C.Biophys J. 2021 Jul 20;120(14):2927-2942. doi: 10.1016/j.bpj.2021.02.039. Epub 2021 Mar 4. Biophys J. 2021. PMID: 33675766 Free PMC article. Review.
-
Effectiveness of Ultraviolet-C Light and a High-Level Disinfection Cabinet for Decontamination of N95 Respirators.Pathog Immun. 2020 Apr 20;5(1):52-67. doi: 10.20411/pai.v5i1.372. eCollection 2020. Pathog Immun. 2020. PMID: 32363254 Free PMC article.
-
Current Understanding of Ultraviolet-C Decontamination of N95 Filtering Facepiece Respirators.Appl Biosaf. 2021 Jun 1;26(2):90-102. doi: 10.1089/apb.20.0051. Epub 2021 Jun 2. Appl Biosaf. 2021. PMID: 36034687 Free PMC article. Review.
Cited by
-
Ultraviolet-C Irradiation, Heat, and Storage as Potential Methods of Inactivating SARS-CoV-2 and Bacterial Pathogens on Filtering Facepiece Respirators.Pathogens. 2022 Jan 10;11(1):83. doi: 10.3390/pathogens11010083. Pathogens. 2022. PMID: 35056031 Free PMC article.
-
Polymer Additives to Personal Protective Equipment can Inactivate Pathogens.Ann Biomed Eng. 2023 Apr;51(4):833-845. doi: 10.1007/s10439-022-03100-1. Epub 2022 Oct 15. Ann Biomed Eng. 2023. PMID: 36243778 Free PMC article.
References
-
- CDC. Implementing filtering facepiece respirator (FFR) reuse, including reuse after decontamination, when there are known shortages of N95 respirators.: Centers for Disease Control Prevention; 2020. [updated August 4, 2020. May 6, 2021]. Available from: https://www.cdc.gov/coronavirus/2019-ncov/hcp/ppe-strategy/decontaminati....
-
- FDA. Decontamination System EUAs for Personal Protective Equipment: US Food and Drug Administration; 2021. [cited 2021 May 6, 2021]. Available from: https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-em....
-
- Degesys NF, Wang RC, Kwan E, Fahimi J, Noble JA, Raven MC. Correlation Between N95 Extended Use and Reuse and Fit Failure in an Emergency Department. JAMA. 2020;324(1):94–6. doi: 10.1001/jama.2020.9843. PubMed PMID: 32496504; PMCID: PMC7273312. - DOI - PMC - PubMed
-
- Fabre V, Cosgrove SE, Hsu YJ, Jones GF, Helsel T, Bukowski J, Sobota M, Sick-Samuels AC, Milstone AM, Maragakis LL, Rock C, Centers for Disease C, Prevention Epicenters P. N95 filtering face piece respirators remain effective after extensive reuse during the coronavirus disease 2019 (COVID-19) pandemic. Infect Control Hosp Epidemiol. 2021:1–4. doi: 10.1017/ice.2021.76. PubMed PMID: 33602376; PMCID: PMC7971774. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
