Randomized Controlled Trial of Neurokinin 3 Receptor Antagonist Fezolinetant for Treatment of Polycystic Ovary Syndrome
- PMID: 34000049
- PMCID: PMC8372662
- DOI: 10.1210/clinem/dgab320
Randomized Controlled Trial of Neurokinin 3 Receptor Antagonist Fezolinetant for Treatment of Polycystic Ovary Syndrome
Abstract
Context: Polycystic ovary syndrome (PCOS), a highly prevalent endocrine disorder characterized by hyperandrogenism, is the leading cause of anovulatory infertility.
Objective: This proof-of-concept study evaluated clinical efficacy and safety of the neurokinin 3 (NK3) receptor antagonist fezolinetant in PCOS.
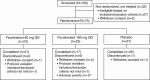
Methods: This was a phase 2a, randomized, double-blind, placebo-controlled, multicenter study (EudraCT 2014-004409-34). The study was conducted at 5 European clinical centers. Women with PCOS participated in the study. Interventions included fezolinetant 60 or 180 mg/day or placebo for 12 weeks. The primary efficacy end point was change in total testosterone. Gonadotropins, ovarian hormones, safety and tolerability were also assessed.
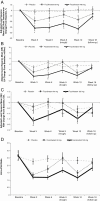
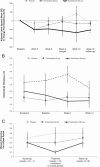
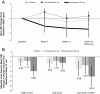
Results: Seventy-three women were randomly assigned, and 64 participants completed the study. Adjusted mean (SE) changes in total testosterone from baseline to week 12 for fezolinetant 180 and 60 mg/day were -0.80 (0.13) and -0.39 (0.12) nmol/L vs -0.05 (0.10) nmol/L with placebo (P < .001 and P < .05, respectively). Adjusted mean (SE) changes from baseline in luteinizing hormone (LH) for fezolinetant 180 and 60 mg/d were -10.17 (1.28) and -8.21 (1.18) vs -3.16 (1.04) IU/L with placebo (P < .001 and P = .002); corresponding changes in follicle-stimulating hormone (FSH) were -1.46 (0.32) and -0.92 (0.30) vs -0.57 (0.26) IU/L (P = .03 and P = .38), underpinning a dose-dependent decrease in the LH-to-FSH ratio vs placebo (P < .001). Circulating levels of progesterone and estradiol did not change significantly vs placebo (P > .10). Fezolinetant was well tolerated.
Conclusion: Fezolinetant had a sustained effect to suppress hyperandrogenism and reduce the LH-to-FSH ratio in women with PCOS.
Keywords: dynorphin A neurons; gonadotropin-releasing hormone; kisspeptin; neurokinin 3 receptor; neurokinin B; polycystic ovary syndrome.
© The Author(s) 2021. Published by Oxford University Press on behalf of the Endocrine Society.
Figures
Comment in
-
Targeting Elevated GnRH Pulsatility to Treat Polycystic Ovary Syndrome.J Clin Endocrinol Metab. 2021 Sep 27;106(10):e4275-e4277. doi: 10.1210/clinem/dgab422. J Clin Endocrinol Metab. 2021. PMID: 34117885 Free PMC article. No abstract available.
Similar articles
-
Treatment of Menopausal Vasomotor Symptoms With Fezolinetant, a Neurokinin 3 Receptor Antagonist: A Phase 2a Trial.J Clin Endocrinol Metab. 2019 Dec 1;104(12):5893-5905. doi: 10.1210/jc.2019-00677. J Clin Endocrinol Metab. 2019. PMID: 31415087 Clinical Trial.
-
Effects of neurokinin 3 receptor antagonist fezolinetant on hot flash-like symptoms in ovariectomized rats.Eur J Pharmacol. 2021 Aug 15;905:174207. doi: 10.1016/j.ejphar.2021.174207. Epub 2021 May 25. Eur J Pharmacol. 2021. PMID: 34048742
-
Neurokinin B Receptor Antagonism in Women With Polycystic Ovary Syndrome: A Randomized, Placebo-Controlled Trial.J Clin Endocrinol Metab. 2016 Nov;101(11):4313-4321. doi: 10.1210/jc.2016-1202. Epub 2016 Jul 26. J Clin Endocrinol Metab. 2016. PMID: 27459523 Clinical Trial.
-
Treatments targeting neuroendocrine dysfunction in polycystic ovary syndrome (PCOS).Clin Endocrinol (Oxf). 2022 Aug;97(2):156-164. doi: 10.1111/cen.14704. Epub 2022 Mar 8. Clin Endocrinol (Oxf). 2022. PMID: 35262967 Review.
-
Neuroendocrine Determinants of Polycystic Ovary Syndrome.Int J Environ Res Public Health. 2022 Mar 6;19(5):3089. doi: 10.3390/ijerph19053089. Int J Environ Res Public Health. 2022. PMID: 35270780 Free PMC article. Review.
Cited by
-
The role of gonadotropin-releasing hormone neurons in polycystic ovary syndrome.J Neuroendocrinol. 2022 May;34(5):e13093. doi: 10.1111/jne.13093. Epub 2022 Jan 26. J Neuroendocrinol. 2022. PMID: 35083794 Free PMC article. Review.
-
Revolutionizing Infertility Management through Novel Peptide-based Targets.Curr Protein Pept Sci. 2024;25(9):738-752. doi: 10.2174/0113892037304433240430144106. Curr Protein Pept Sci. 2024. PMID: 38778605 Review.
-
Available Treatments and Adjunctive Therapies for Polycystic Ovarian Syndrome (PCOS) Patients of Reproductive Age: A Scoping Review.Cureus. 2024 Sep 30;16(9):e70501. doi: 10.7759/cureus.70501. eCollection 2024 Sep. Cureus. 2024. PMID: 39479136 Free PMC article.
-
FDA approved fezolinetant (Veozah): a critical evaluation of its efficacy and safety for menopausal vasomotor symptoms, calling for prospective research.Arch Womens Ment Health. 2024 Dec;27(6):943-946. doi: 10.1007/s00737-024-01456-y. Epub 2024 Mar 13. Arch Womens Ment Health. 2024. PMID: 38478035
-
Targeting Elevated GnRH Pulsatility to Treat Polycystic Ovary Syndrome.J Clin Endocrinol Metab. 2021 Sep 27;106(10):e4275-e4277. doi: 10.1210/clinem/dgab422. J Clin Endocrinol Metab. 2021. PMID: 34117885 Free PMC article. No abstract available.
References
-
- Duncan WC. A guide to understanding polycystic ovary syndrome (PCOS). J Fam Plann Reprod Health Care. 2014;40(3):217-225. - PubMed
-
- Bozdag G, Mumusoglu S, Zengin D, Karabulut E, Yildiz BO. The prevalence and phenotypic features of polycystic ovary syndrome: a systematic review and meta-analysis. Hum Reprod. 2016;31(12):2841-2855. - PubMed
-
- Azziz R, Marin C, Hoq L, Badamgarav E, Song P. Health care-related economic burden of the polycystic ovary syndrome during the reproductive life span. J Clin Endocrinol Metab. 2005;90(8):4650-4658. - PubMed
-
- Jason J. Polycystic ovary syndrome in the United States: clinical visit rates, characteristics, and associated health care costs. Arch Intern Med. 2011;171(13):1209-1211. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

