Evaluation of an oxygen-dependent self-inducible surfactin synthesis in B. subtilis by substitution of native promoter PsrfA by anaerobically active PnarG and PnasD
- PMID: 33876328
- PMCID: PMC8055807
- DOI: 10.1186/s13568-021-01218-4
Evaluation of an oxygen-dependent self-inducible surfactin synthesis in B. subtilis by substitution of native promoter PsrfA by anaerobically active PnarG and PnasD
Abstract
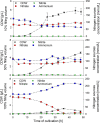
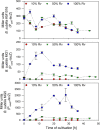
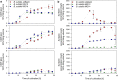
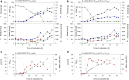
A novel approach targeting self-inducible surfactin synthesis under oxygen-limited conditions is presented. Because both the nitrate (NarGHI) and nitrite (NasDE) reductase are highly expressed during anaerobic growth of B. subtilis, the native promoter PsrfA of the surfactin operon in strain B. subtilis JABs24 was replaced by promoters PnarG and PnasD to induce surfactin synthesis anaerobically. Shake flask cultivations with varying oxygen availabilities indicated no significant differences in native PsrfA expression. As hypothesized, activity of PnarG and PnasD increased with lower oxygen levels and surfactin was not produced by PsrfA::PnarG as well as PsrfA::PnasD mutant strains under conditions with highest oxygen availability. PnarG showed expressions similar to PsrfA at lowest oxygen availability, while maximum value of PnasD was more than 5.5-fold higher. Although the promoter exchange PsrfA::PnarG resulted in a decreased surfactin titer at lowest oxygen availability, the strain carrying PsrfA::PnasD reached a 1.4-fold increased surfactin concentration with 696 mg/L and revealed an exceptional high overall YP/X of 1.007 g/g. This value also surpassed the YP/X of the reference strain JABs24 at highest and moderate oxygen availability. Bioreactor cultivations illustrated that significant cell lysis occurred when the process of "anaerobization" was performed too fast. However, processes with a constantly low agitation and aeration rate showed promising potential for process improvement, especially by employing the strain carrying PsrfA::PnasD promoter exchange. Additionally, replacement of other native promoters by nitrite reductase promoter PnasD represents a promising tool for anaerobic-inducible bioprocesses in Bacillus.
Keywords: Bacillus subtilis; Lipopeptide biosurfactants; Microaerobic; Oxygen; Promoter exchange; Surfactin.
Conflict of interest statement
The authors declare that they have no conflict of interests.
Figures
Similar articles
-
In situ enhancement of surfactin biosynthesis in Bacillus subtilis using novel artificial inducible promoters.Biotechnol Bioeng. 2017 Apr;114(4):832-842. doi: 10.1002/bit.26197. Epub 2016 Oct 21. Biotechnol Bioeng. 2017. PMID: 27723092
-
Influence of B. subtilis 3NA mutations in spo0A and abrB on surfactin production in B. subtilis 168.Microb Cell Fact. 2021 Sep 26;20(1):188. doi: 10.1186/s12934-021-01679-z. Microb Cell Fact. 2021. PMID: 34565366 Free PMC article.
-
Towards the Anaerobic Production of Surfactin Using Bacillus subtilis.Front Bioeng Biotechnol. 2020 Nov 26;8:554903. doi: 10.3389/fbioe.2020.554903. eCollection 2020. Front Bioeng Biotechnol. 2020. PMID: 33324620 Free PMC article.
-
Bacillus licheniformis: The unexplored alternative for the anaerobic production of lipopeptide biosurfactants?Biotechnol Adv. 2022 Nov;60:108013. doi: 10.1016/j.biotechadv.2022.108013. Epub 2022 Jun 22. Biotechnol Adv. 2022. PMID: 35752271 Review.
-
Molecular genetics of surfactin and its effects on different sub-populations of Bacillus subtilis.Biotechnol Rep (Amst). 2021 Oct 26;32:e00686. doi: 10.1016/j.btre.2021.e00686. eCollection 2021 Dec. Biotechnol Rep (Amst). 2021. PMID: 34786355 Free PMC article. Review.
Cited by
-
Riboswitch-mediated regulation of riboflavin biosynthesis genes in prokaryotes.3 Biotech. 2022 Oct;12(10):278. doi: 10.1007/s13205-022-03348-3. Epub 2022 Sep 14. 3 Biotech. 2022. PMID: 36275359 Free PMC article. Review.
-
Expression of degQ gene and its effect on lipopeptide production as well as formation of secretory proteases in Bacillus subtilis strains.Microbiologyopen. 2021 Oct;10(5):e1241. doi: 10.1002/mbo3.1241. Microbiologyopen. 2021. PMID: 34713601 Free PMC article.
-
Modeling the time course of ComX: towards molecular process control for Bacillus wild-type cultivations.AMB Express. 2021 Oct 29;11(1):144. doi: 10.1186/s13568-021-01306-5. AMB Express. 2021. PMID: 34714452 Free PMC article.
-
Characterization ofantifungal properties of lipopeptide-producing Bacillus velezensis strains and their proteome-based response to the phytopathogens, Diaporthe spp.Front Bioeng Biotechnol. 2023 Aug 7;11:1228386. doi: 10.3389/fbioe.2023.1228386. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 37609113 Free PMC article.
-
The phylogeny and metabolic potentials of an n-alkane-degrading Venatorbacter bacterium isolated from deep-sea sediment of the Mariana Trench.Front Microbiol. 2023 Mar 22;14:1108651. doi: 10.3389/fmicb.2023.1108651. eCollection 2023. Front Microbiol. 2023. PMID: 37032874 Free PMC article.
References
-
- Chtioui O, Dimitrov K, Gancel F, Dhulster P, Nikov J. Rotating discs bioreactor, a new tool for lipopeptides production. Process Biochem. 2012;5:2020. doi: 10.1016/j.procbio.2012.07.013. - DOI
-
- Coutte F, Leclère V, Béchet M, Guez J-S, Lecouturier D, Chollet-Imbert M, Kessler D, et al. Effect of pps disruption and constitutive expression of srfA on surfactin productivity, spreading and antagonistic properties of Bacillus subtilis 168 derivatives. J Appl Microbiol. 2010;109:480–491. - PubMed
-
- Davis DA, Lynch HC, Varley J. The production of Surfactin in batch culture by Bacillus subtilis ATCC 21332 is strongly influenced by the conditions of nitrogen metabolism. Enzyme Microbial Technol. 1999;25(3–5):322–329. doi: 10.1016/S0141-0229(99)00048-4. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

