Complement receptor 3 mediates both sinking phagocytosis and phagocytic cup formation via distinct mechanisms
- PMID: 33839682
- PMCID: PMC7948798
- DOI: 10.1016/j.jbc.2021.100256
Complement receptor 3 mediates both sinking phagocytosis and phagocytic cup formation via distinct mechanisms
Abstract
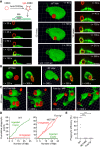
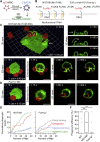
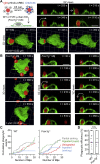
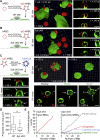
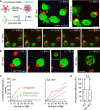
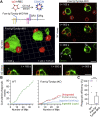
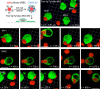
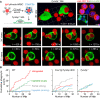
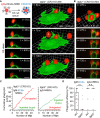
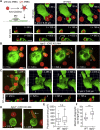
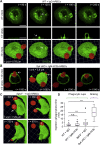
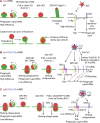
A long-standing hypothesis is that complement receptors (CRs), especially CR3, mediate sinking phagocytosis, but evidence is lacking. Alternatively, CRs have been reported to induce membrane ruffles or phagocytic cups, akin to those induced by Fcγ receptors (FcγRs), but the details of these events are unclear. Here we used real-time 3D imaging and KO mouse models to clarify how particles (human red blood cells) are internalized by resident peritoneal F4/80+ cells (macrophages) via CRs and/or FcγRs. We first show that FcγRs mediate highly efficient, rapid (2-3 min) phagocytic cup formation, which is completely abolished by deletion or mutation of the FcR γ chain or conditional deletion of the signal transducer Syk. FcγR-mediated phagocytic cups robustly arise from any point of cell-particle contact, including filopodia. In the absence of CR3, FcγR-mediated phagocytic cups exhibit delayed closure and become aberrantly elongated. Independent of FcγRs, CR3 mediates sporadic ingestion of complement-opsonized particles by rapid phagocytic cup-like structures, typically emanating from membrane ruffles and largely prevented by deletion of the immunoreceptor tyrosine-based activation motif (ITAM) adaptors FcR γ chain and DAP12 or Syk. Deletion of ITAM adaptors or Syk clearly revealed that there is a slow (10-25 min) sinking mode of phagocytosis via a restricted orifice. In summary, we show that (1) CR3 indeed mediates a slow sinking mode of phagocytosis, which is accentuated by deletion of ITAM adaptors or Syk, (2) CR3 induces phagocytic cup-like structures, driven by ITAM adaptors and Syk, and (3) CR3 is involved in forming and closing FcγR-mediated phagocytic cups.
Keywords: Complement system; knockout mice; live-cell imaging; macrophages; phagocytosis.
Copyright © 2021 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interest The authors declare that they have no conflicts of interest with the contents of this article.
Figures
Similar articles
-
Complement receptor 3 mediates both sinking phagocytosis and phagocytic cup formation via distinct mechanisms.J Biol Chem. 2021 Jan 4:jbc.RA120.015346. doi: 10.1074/jbc.RA120.015346. Online ahead of print. J Biol Chem. 2021. PMID: 33397789
-
Regulation of FcgammaR-stimulated phagocytosis by the 72-kDa inositol polyphosphate 5-phosphatase: SHIP1, but not the 72-kDa 5-phosphatase, regulates complement receptor 3 mediated phagocytosis by differential recruitment of these 5-phosphatases to the phagocytic cup.Blood. 2007 Dec 15;110(13):4480-91. doi: 10.1182/blood-2007-02-073874. Epub 2007 Aug 6. Blood. 2007. PMID: 17682126
-
CR3 ruffles FcγR's claim over phagocytic cups.J Biol Chem. 2021 Jan-Jun;296:100801. doi: 10.1016/j.jbc.2021.100801. Epub 2021 May 19. J Biol Chem. 2021. PMID: 34019878 Free PMC article.
-
Tyrosine phosphorylation and Fcgamma receptor-mediated phagocytosis.FEBS Lett. 1997 Jan 2;400(1):11-4. doi: 10.1016/s0014-5793(96)01359-2. FEBS Lett. 1997. PMID: 9000504 Review.
-
Protein tyrosine kinase, syk: a key player in phagocytic cells.J Biochem. 2009 Mar;145(3):267-73. doi: 10.1093/jb/mvp001. Epub 2009 Jan 4. J Biochem. 2009. PMID: 19124456 Review.
Cited by
-
Cellular Uptake of Phase-Separating Peptide Coacervates.Adv Sci (Weinh). 2024 Nov;11(42):e2402652. doi: 10.1002/advs.202402652. Epub 2024 Aug 30. Adv Sci (Weinh). 2024. PMID: 39214144 Free PMC article.
-
Navigating Q fever: Current perspectives and challenges in outbreak preparedness.Open Vet J. 2024 Oct;14(10):2509-2524. doi: 10.5455/OVJ.2024.v14.i10.2. Epub 2024 Oct 31. Open Vet J. 2024. PMID: 39545195 Free PMC article. Review.
-
Phagocytic 'teeth' and myosin-II 'jaw' power target constriction during phagocytosis.Elife. 2021 Oct 28;10:e68627. doi: 10.7554/eLife.68627. Elife. 2021. PMID: 34708690 Free PMC article.
-
Building the phagocytic cup on an actin scaffold.Curr Opin Cell Biol. 2022 Aug;77:102112. doi: 10.1016/j.ceb.2022.102112. Epub 2022 Jul 9. Curr Opin Cell Biol. 2022. PMID: 35820329 Free PMC article. Review.
-
Monocytes, particularly nonclassical ones, lose their opsonic and nonopsonic phagocytosis capacity during pediatric cerebral malaria.Front Immunol. 2024 May 21;15:1358853. doi: 10.3389/fimmu.2024.1358853. eCollection 2024. Front Immunol. 2024. PMID: 38835780 Free PMC article.
References
-
- Tauber A.I. Metchnikoff and the phagocytosis theory. Nat. Rev. Mol. Cell Biol. 2003;4:897–901. - PubMed
-
- Flannagan R.S., Jaumouille V., Grinstein S. The cell biology of phagocytosis. Annu. Rev. Pathol. 2012;7:61–98. - PubMed
-
- Munthe-Kaas A.C., Kaplan G., Seljelid R. On the mechanism of internalization of opsonized particles by rat Kupffer cells in vitro. Exp. Cell Res. 1976;103:201–212. - PubMed
-
- Kaplan G. Differences in the mode of phagocytosis with Fc and C3 receptors in macrophages. Scand. J. Immunol. 1977;6:797–807. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

