Interplay of mesoscale physics and agent-like behaviors in the parallel evolution of aggregative multicellularity
- PMID: 33062243
- PMCID: PMC7549232
- DOI: 10.1186/s13227-020-00165-8
Interplay of mesoscale physics and agent-like behaviors in the parallel evolution of aggregative multicellularity
Abstract
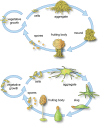
Myxobacteria and dictyostelids are prokaryotic and eukaryotic multicellular lineages, respectively, that after nutrient depletion aggregate and develop into structures called fruiting bodies. The developmental processes and resulting morphological outcomes resemble one another to a remarkable extent despite their independent origins, the evolutionary distance between them and the lack of traceable homology in molecular mechanisms. We hypothesize that the morphological parallelism between the two lineages arises as the consequence of the interplay within multicellular aggregates between generic processes, physical and physicochemical processes operating similarly in living and non-living matter at the mesoscale (~10-3-10-1 m) and agent-like behaviors, unique to living systems and characteristic of the constituent cells, considered as autonomous entities acting according to internal rules in a shared environment. Here, we analyze the contributions of generic and agent-like determinants in myxobacteria and dictyostelid development and their roles in the generation of their common traits. Consequent to aggregation, collective cell-cell contacts mediate the emergence of liquid-like properties, making nascent multicellular masses subject to novel patterning and morphogenetic processes. In both lineages, this leads to behaviors such as streaming, rippling, and rounding-up, as seen in non-living fluids. Later the aggregates solidify, leading them to exhibit additional generic properties and motifs. Computational models suggest that the morphological phenotypes of the multicellular masses deviate from the predictions of generic physics due to the contribution of agent-like behaviors of cells such as directed migration, quiescence, and oscillatory signal transduction mediated by responses to external cues. These employ signaling mechanisms that reflect the evolutionary histories of the respective organisms. We propose that the similar developmental trajectories of myxobacteria and dictyostelids are more due to shared generic physical processes in coordination with analogous agent-type behaviors than to convergent evolution under parallel selection regimes. Insights from the biology of these aggregative forms may enable a unified understanding of developmental evolution, including that of animals and plants.
Keywords: Deformable solids; Dictyostelids; Excitable media; Liquid tissues; Myxobacteria.
© The Author(s) 2020.
Conflict of interest statement
Competing interestsThe authors declare no competing interests.
Figures

Similar articles
-
An Evo-Devo Perspective on Multicellular Development of Myxobacteria.J Exp Zool B Mol Dev Evol. 2017 Jan;328(1-2):165-178. doi: 10.1002/jez.b.22727. J Exp Zool B Mol Dev Evol. 2017. PMID: 28217903 Review.
-
Dynamical patterning modules and network motifs as joint determinants of development: Lessons from an aggregative bacterium.J Exp Zool B Mol Dev Evol. 2021 Apr;336(3):300-314. doi: 10.1002/jez.b.22946. Epub 2020 May 17. J Exp Zool B Mol Dev Evol. 2021. PMID: 32419346
-
Why have aggregative multicellular organisms stayed simple?Curr Genet. 2021 Dec;67(6):871-876. doi: 10.1007/s00294-021-01193-0. Epub 2021 Jun 10. Curr Genet. 2021. PMID: 34114051 Review.
-
Two potential evolutionary origins of the fruiting bodies of the dictyostelid slime moulds.Biol Rev Camb Philos Soc. 2019 Oct;94(5):1591-1604. doi: 10.1111/brv.12516. Epub 2019 Apr 15. Biol Rev Camb Philos Soc. 2019. PMID: 30989827 Review.
-
Dynamical patterning modules: physico-genetic determinants of morphological development and evolution.Phys Biol. 2008 Apr 10;5(1):015008. doi: 10.1088/1478-3975/5/1/015008. Phys Biol. 2008. PMID: 18403826
Cited by
-
A Cellular Potts Model of the interplay of synchronization and aggregation.PeerJ. 2024 Feb 29;12:e16974. doi: 10.7717/peerj.16974. eCollection 2024. PeerJ. 2024. PMID: 38435996 Free PMC article.
-
Group-selection via aggregative propagule-formation enables cooperative multicellularity in an individual based, spatial model.PLoS Comput Biol. 2024 May 7;20(5):e1012107. doi: 10.1371/journal.pcbi.1012107. eCollection 2024 May. PLoS Comput Biol. 2024. PMID: 38713735 Free PMC article.
-
Single-cell phenotypic plasticity modulates social behavior in Dictyostelium discoideum.iScience. 2023 May 2;26(6):106783. doi: 10.1016/j.isci.2023.106783. eCollection 2023 Jun 16. iScience. 2023. PMID: 37235054 Free PMC article.
-
Unicellular-multicellular evolutionary branching driven by resource limitations.J R Soc Interface. 2022 Jun;19(191):20220018. doi: 10.1098/rsif.2022.0018. Epub 2022 Jun 1. J R Soc Interface. 2022. PMID: 35642429 Free PMC article.
-
The Shared Origins of Embodiment and Development.Front Syst Neurosci. 2021 Aug 17;15:726403. doi: 10.3389/fnsys.2021.726403. eCollection 2021. Front Syst Neurosci. 2021. PMID: 34483853 Free PMC article.
References
-
- Maynard Smith J, Szathmáry E. The major transitions in evolution. Oxford: W.H. Freeman Spektrum; 1995.
-
- Niklas KJ, Newman SA, editors. Multicellularity: origins and evolution. Cambridge: The MIT Press; 2016.
Publication types
LinkOut - more resources
Full Text Sources

