Effect of setmelanotide, a melanocortin-4 receptor agonist, on obesity in Bardet-Biedl syndrome
- PMID: 32627316
- PMCID: PMC7689750
- DOI: 10.1111/dom.14133
Effect of setmelanotide, a melanocortin-4 receptor agonist, on obesity in Bardet-Biedl syndrome
Abstract
Aim: To report an analysis of ~1 year of setmelanotide treatment for obesity and hunger, as well as metabolic and cardiac outcomes, in individuals with Bardet-Biedl syndrome (BBS).
Materials and methods: Individuals aged 12 years and older with BBS received once-daily setmelanotide. The dose was titrated every 2 weeks to establish the individual therapeutic dose (≤3 mg); treatment continued for an additional 10 weeks. Participants who lost 5 kg or more (or ≥5% of body weight if <100 kg at baseline) continued into the 52-week extension phase. The primary outcome was mean percent change from baseline in body weight at 3 months. Hunger scores and safety were secondary outcomes.
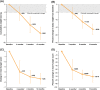
Results: From February 2017 and February 2018, 10 individuals were screened; eight completed the 3-month treatment phase and seven completed the extension phase. Mean percent change in body weight from baseline to 3 months was -5.5% (90% CI, -9.3% to -1.6%; n = 8); change from baseline was -11.3% (90% CI, -15.5% to -7.0%; n = 8) at 6 months and -16.3% (90% CI, -19.9% to -12.8%; n = 7) at 12 months. All participants reported at least one treatment-emergent adverse event (AE), most commonly injection-site reaction. No AEs led to study withdrawal or death. Most, morning, and average hunger scores were reduced across time points.
Conclusions: Setmelanotide reduced body weight and hunger in individuals with BBS and had a safety profile consistent with previous reports. Setmelanotide may be a treatment option in individuals with BBS-associated obesity and hyperphagia.
Keywords: antiobesity drug, appetite control, obesity therapy, phase I-II study.
© 2020 The Authors. Diabetes, Obesity and Metabolism published by John Wiley & Sons Ltd.
Conflict of interest statement
RH is a consultant for Rhythm Pharmaceuticals, Inc., and Trinity Life Sciences. He receives grant funding from the Bardet‐Biedl Syndrome Foundation. JY receives grant support for clinical investigations from the NICHD, NIH, Soleno Pharmaceuticals Inc., and Rhythm Pharmaceuticals, Inc. GY, GG and MS are employed by and may own stock in Rhythm Pharmaceuticals, Inc. KF, ED and SB have no conflicts of interest to disclose.
Figures
Similar articles
-
Efficacy and safety of setmelanotide, a melanocortin-4 receptor agonist, in patients with Bardet-Biedl syndrome and Alström syndrome: a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial with an open-label period.Lancet Diabetes Endocrinol. 2022 Dec;10(12):859-868. doi: 10.1016/S2213-8587(22)00277-7. Epub 2022 Nov 7. Lancet Diabetes Endocrinol. 2022. PMID: 36356613 Free PMC article. Clinical Trial.
-
An evaluation of setmelanotide injection for chronic weight management in adult and pediatric patients with obesity due to Bardet-Biedl syndrome.Expert Opin Pharmacother. 2023 Apr;24(6):667-674. doi: 10.1080/14656566.2023.2199152. Epub 2023 Apr 6. Expert Opin Pharmacother. 2023. PMID: 37013719 Free PMC article.
-
Efficacy and safety of setmelanotide, an MC4R agonist, in individuals with severe obesity due to LEPR or POMC deficiency: single-arm, open-label, multicentre, phase 3 trials.Lancet Diabetes Endocrinol. 2020 Dec;8(12):960-970. doi: 10.1016/S2213-8587(20)30364-8. Epub 2020 Oct 30. Lancet Diabetes Endocrinol. 2020. PMID: 33137293 Clinical Trial.
-
Setmelanotide: First Approval.Drugs. 2021 Feb;81(3):397-403. doi: 10.1007/s40265-021-01470-9. Drugs. 2021. PMID: 33638809 Review.
-
[Improved Care and Treatment Options for Patients with Hyperphagia-Associated Obesity in Bardet-Biedl Syndrome].Klin Padiatr. 2024 Sep;236(5):269-279. doi: 10.1055/a-2251-5382. Epub 2024 Mar 8. Klin Padiatr. 2024. PMID: 38458231 Free PMC article. Review. German.
Cited by
-
Bardet-Biedl Syndrome: Current Perspectives and Clinical Outlook.Ther Clin Risk Manag. 2023 Jan 30;19:115-132. doi: 10.2147/TCRM.S338653. eCollection 2023. Ther Clin Risk Manag. 2023. PMID: 36741589 Free PMC article. Review.
-
Setmelanotide: A Melanocortin-4 Receptor Agonist for the Treatment of Severe Obesity Due to Hypothalamic Dysfunction.touchREV Endocrinol. 2024 Oct;20(2):62-71. doi: 10.17925/EE.2024.20.2.9. Epub 2024 Feb 9. touchREV Endocrinol. 2024. PMID: 39526054 Free PMC article. Review.
-
Genetic obesity: an update with emerging therapeutic approaches.Ann Pediatr Endocrinol Metab. 2022 Sep;27(3):169-175. doi: 10.6065/apem.2244188.094. Epub 2022 Sep 30. Ann Pediatr Endocrinol Metab. 2022. PMID: 36203267 Free PMC article.
-
Rare genetic forms of obesity in childhood and adolescence: A narrative review of the main treatment options with a focus on innovative pharmacological therapies.Eur J Pediatr. 2024 Apr;183(4):1499-1508. doi: 10.1007/s00431-024-05427-4. Epub 2024 Jan 16. Eur J Pediatr. 2024. PMID: 38227053 Review.
-
Risk Factors, Clinical Consequences, Prevention, and Treatment of Childhood Obesity.Children (Basel). 2022 Dec 16;9(12):1975. doi: 10.3390/children9121975. Children (Basel). 2022. PMID: 36553418 Free PMC article. Review.
References
-
- Geets E, Meuwissen MEC, Van Hul W. Clinical, molecular genetics and therapeutic aspects of syndromic obesity. Clin Genet. 2019;95(1):23‐40. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

