Coinfection rates and clinical outcome data for cytomegalovirus and Epstein-Barr virus in post-transplant patients: A systematic review of the literature
- PMID: 32603496
- PMCID: PMC7816247
- DOI: 10.1111/tid.13396
Coinfection rates and clinical outcome data for cytomegalovirus and Epstein-Barr virus in post-transplant patients: A systematic review of the literature
Abstract
Background: In transplant recipients, cytomegalovirus (CMV) infection increases morbidity and mortality; furthermore, coinfection with other human herpesviruses like the Epstein-Barr virus (EBV) may complicate their management. This systematic literature review aimed to summarize rates of CMV-EBV coinfection and associated clinical outcomes among solid organ transplant (SOT) and hematopoietic stem cell transplant (HSCT) recipients.
Methods: An electronic literature search was performed using pre-specified search strategies (January 1, 2010-October 31, 2018) and following established/best practice methodology. Of 316 publications identified, 294 did not report CMV-EBV coinfection and were excluded. Studies meeting the inclusion criteria were further analyzed. Due to limited reporting/heterogeneity, data were not meta-analyzable.
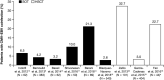
Results: Nine studies (six SOT; three HSCT) reported CMV-EBV coinfection; rates of coinfection post transplantation varied between 2.6% and 32.7%. Two studies indicated CMV reactivation to be an independent variable associated with EBV reactivation. Among SOT studies, higher rates of graft dysfunction (47.4% vs 22.9%), rejection episodes (20.0% vs 8.9%), or acute rejection (50.0% vs 31.0%) were reported for patients with coinfection than without. In HSCT studies, patients with graft-vs-host disease were not reported separately for coinfection. Two studies described cases of post-transplant lymphoproliferative disorder (PTLD) in patients with CMV-EBV coinfection and reported rates of PTLD of 92% and 100%.
Conclusion: The CMV-EBV coinfection rate in HSCT and SOT recipients varied and was associated with increased graft rejection and PTLD compared with patients without coinfection. Further research may improve understanding of the burden of CMV-EBV coinfection among transplant recipients.
Keywords: Epstein-Barr virus; cytomegalovirus; hematopoietic stem cell transplant; solid organ transplant.
© 2020 The Authors. Transplant Infectious Disease published by Wiley Periodicals LLC.
Conflict of interest statement
CA‐S and IH are employees of, and hold stock in, Shire International GmbH, a Takeda company. ERB is an employee of CTI Clinical Trial and Consulting Services, which was funded by Shire, a Takeda company, to conduct this study.
Figures
Similar articles
-
Co-infection of Cytomegalovirus and Epstein-Barr Virus Diminishes the Frequency of CD56dimNKG2A+KIR- NK Cells and Contributes to Suboptimal Control of EBV in Immunosuppressed Children With Post-transplant Lymphoproliferative Disorder.Front Immunol. 2020 Jun 17;11:1231. doi: 10.3389/fimmu.2020.01231. eCollection 2020. Front Immunol. 2020. PMID: 32625211 Free PMC article.
-
[Clinical Study of Allogeneic Hematopoietic Stem Cell Transplantation Patients with Co-Reactivation of Cytomegalovirus and Epstein-Barr Virus].Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2024 Apr;32(2):561-567. doi: 10.19746/j.cnki.issn.1009-2137.2024.02.036. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2024. PMID: 38660867 Chinese.
-
Co-Reactivation of Cytomegalovirus and Epstein-Barr Virus Was Associated With Poor Prognosis After Allogeneic Stem Cell Transplantation.Front Immunol. 2021 Feb 16;11:620891. doi: 10.3389/fimmu.2020.620891. eCollection 2020. Front Immunol. 2021. PMID: 33664733 Free PMC article.
-
Epstein-Barr virus-related post-transplant lymphoproliferative disorder in solid organ transplant recipients.Clin Microbiol Infect. 2014 Sep;20 Suppl 7:109-18. doi: 10.1111/1469-0691.12534. Clin Microbiol Infect. 2014. PMID: 24475976 Review.
-
Outcome of treatment of Epstein-Barr virus-related post-transplant lymphoproliferative disorder in hematopoietic stem cell recipients: a comprehensive review of reported cases.Transpl Infect Dis. 2009 Oct;11(5):383-92. doi: 10.1111/j.1399-3062.2009.00411.x. Epub 2009 Jun 23. Transpl Infect Dis. 2009. PMID: 19558376 Review.
Cited by
-
The human cytomegalovirus decathlon: Ten critical replication events provide opportunities for restriction.Front Cell Dev Biol. 2022 Nov 25;10:1053139. doi: 10.3389/fcell.2022.1053139. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 36506089 Free PMC article. Review.
-
The Contribution of Human Herpes Viruses to γδ T Cell Mobilisation in Co-Infections.Viruses. 2021 Nov 26;13(12):2372. doi: 10.3390/v13122372. Viruses. 2021. PMID: 34960641 Free PMC article. Review.
-
Association Between Cytomegalovirus and Epstein-Barr Virus Co-Reactivation and Hematopoietic Stem Cell Transplantation.Front Cell Infect Microbiol. 2022 Mar 25;12:818167. doi: 10.3389/fcimb.2022.818167. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35402291 Free PMC article.
-
The Role of Coinfections in the EBV-Host Broken Equilibrium.Viruses. 2021 Jul 19;13(7):1399. doi: 10.3390/v13071399. Viruses. 2021. PMID: 34372605 Free PMC article. Review.
-
Longitudinal Monitoring of DNA Viral Loads in Transplant Patients Using Quantitative Metagenomic Next-Generation Sequencing.Pathogens. 2022 Feb 11;11(2):236. doi: 10.3390/pathogens11020236. Pathogens. 2022. PMID: 35215180 Free PMC article.
References
-
- Kotton CN, Fishman JA. Viral infection in the renal transplant recipient. J Am Soc Nephrol. 2005;16(6):1758‐1774. - PubMed
-
- Tolkoff‐Rubin NE, Rubin RH. Control of herpesvirus infection in organ transplant recipients. Can J Infect Dis. 1993;4(suppl C):59C‐64C.

