Combinatorial effects of antibiotics and enzymes against dual-species Staphylococcus aureus and Pseudomonas aeruginosa biofilms in the wound-like medium
- PMID: 32584878
- PMCID: PMC7316268
- DOI: 10.1371/journal.pone.0235093
Combinatorial effects of antibiotics and enzymes against dual-species Staphylococcus aureus and Pseudomonas aeruginosa biofilms in the wound-like medium
Abstract
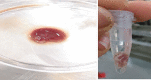
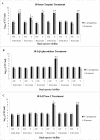
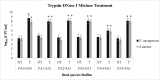
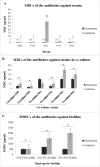
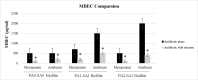
Bacterial biofilms are one of the major issues in the treatment of chronic infections such as chronic wounds, where biofilms are typically polymicrobial. The synergy between species can occur during most polymicrobial infections, where antimicrobial resistance enhances as a result. Furthermore, self-produced extracellular polymeric substance (EPS) in biofilms results in a high tolerance to antibiotics that complicates wound healing. Since most antibiotics fail to remove biofilms in chronic infections, new therapeutic modalities may be required. Disruption of EPS is one of the effective approaches for biofilm eradication. Therefore, degradation of EPS using enzymes may result in improved chronic wounds healing. In the current study, we investigated the efficacy of trypsin, β-glucosidase, and DNase I enzymes on the degradation of dual-species biofilms of Pseudomonas aeruginosa and Staphylococcus aureus in a wound-like medium. These species are the two most common bacteria associated with biofilm formation in chronic wounds. Moreover, the reduction of minimum biofilm eradication concentration (MBEC) of meropenem and amikacin was evaluated when combined with enzymes. The minimum effective concentrations of trypsin, β-glucosidase, and DNase I enzymes to degrade biofilms were 1 μg/ml, 8 U/ml, and 150 U/ml, respectively. Combination of 0.15 μg/ml trypsin and 50 U/ml DNase I had a significant effect on S. aureus-P. aeruginosa biofilms which resulted in the dispersal and dissolution of all biofilms. In the presence of the enzymatic mixture, MBECs of antibiotics showed a significant decrease (p < 0.05), at least 2.5 fold. We found that trypsin/DNase I mixture can be used as an anti-biofilm agent against dual-species biofilms of S. aureus-P. aeruginosa.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
D-amino acids enhance the activity of antimicrobials against biofilms of clinical wound isolates of Staphylococcus aureus and Pseudomonas aeruginosa.Antimicrob Agents Chemother. 2014 Aug;58(8):4353-61. doi: 10.1128/AAC.02468-14. Epub 2014 May 19. Antimicrob Agents Chemother. 2014. PMID: 24841260 Free PMC article.
-
The interplay of Pseudomonas aeruginosa and Staphylococcus aureus in dual-species biofilms impacts development, antibiotic resistance and virulence of biofilms in in vitro wound infection models.PLoS One. 2024 May 28;19(5):e0304491. doi: 10.1371/journal.pone.0304491. eCollection 2024. PLoS One. 2024. PMID: 38805522 Free PMC article.
-
HOCl-producing electrochemical bandage is active in murine polymicrobial wound infection.Microbiol Spectr. 2024 Oct 3;12(10):e0062624. doi: 10.1128/spectrum.00626-24. Epub 2024 Aug 20. Microbiol Spectr. 2024. PMID: 39162542 Free PMC article.
-
Pseudomonas aeruginosa and Staphylococcus aureus communication in biofilm infections: insights through network and database construction.Crit Rev Microbiol. 2019 Sep-Nov;45(5-6):712-728. doi: 10.1080/1040841X.2019.1700209. Epub 2019 Dec 13. Crit Rev Microbiol. 2019. PMID: 31835971 Review.
-
Chronic wound infections: the role of Pseudomonas aeruginosa and Staphylococcus aureus.Expert Rev Anti Infect Ther. 2015 May;13(5):605-13. doi: 10.1586/14787210.2015.1023291. Epub 2015 Mar 8. Expert Rev Anti Infect Ther. 2015. PMID: 25746414 Review.
Cited by
-
Fingolimod Inhibits Exopolysaccharide Production and Regulates Relevant Genes to Eliminate the Biofilm of K. pneumoniae.Int J Mol Sci. 2024 Jan 23;25(3):1397. doi: 10.3390/ijms25031397. Int J Mol Sci. 2024. PMID: 38338675 Free PMC article.
-
Biofilm Formation by Pathogenic Bacteria: Applying a Staphylococcus aureus Model to Appraise Potential Targets for Therapeutic Intervention.Pathogens. 2022 Mar 23;11(4):388. doi: 10.3390/pathogens11040388. Pathogens. 2022. PMID: 35456063 Free PMC article. Review.
-
Advances in the Sensing and Treatment of Wound Biofilms.Angew Chem Weinheim Bergstr Ger. 2022 Mar 21;134(13):e202112218. doi: 10.1002/ange.202112218. Epub 2022 Feb 3. Angew Chem Weinheim Bergstr Ger. 2022. PMID: 38505642 Free PMC article. Review.
-
The activity of hydrolytic enzymes and antibiotics against biofilms of bacteria isolated from industrial-scale cooling towers.Microb Cell Fact. 2024 Oct 16;23(1):282. doi: 10.1186/s12934-024-02502-1. Microb Cell Fact. 2024. PMID: 39415191 Free PMC article.
-
In vitro activities of cellulase and ceftazidime, alone and in combination against Pseudomonas aeruginosa biofilms.BMC Microbiol. 2021 Dec 16;21(1):347. doi: 10.1186/s12866-021-02411-y. BMC Microbiol. 2021. PMID: 34915848 Free PMC article.
References
-
- Burmølle M, Webb JS, Rao D, Hansen LH, Sørensen SJ, Kjelleberg S. Enhanced biofilm formation and increased resistance to antimicrobial agents and bacterial invasion are caused by synergistic interactions in multispecies biofilms. Appl Environ Microbiol. 2006; 72(6):3916–23. 10.1128/AEM.03022-05 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

