Hypoxia-Inducible Factor Activators in Renal Anemia: Current Clinical Experience
- PMID: 31477256
- PMCID: PMC7318915
- DOI: 10.1053/j.ackd.2019.04.004
Hypoxia-Inducible Factor Activators in Renal Anemia: Current Clinical Experience
Abstract
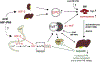
Prolyl hydroxylase domain oxygen sensors are dioxygenases that regulate the activity of hypoxia-inducible factor (HIF), which controls renal and hepatic erythropoietin production and coordinates erythropoiesis with iron metabolism. Small molecule inhibitors of prolyl hydroxylase domain dioxygenases (HIF-PHI [prolyl hydroxylase inhibitor]) stimulate the production of endogenous erythropoietin and improve iron metabolism resulting in efficacious anemia management in patients with CKD. Three oral HIF-PHIs-daprodustat, roxadustat, and vadadustat-have now advanced to global phase III clinical development culminating in the recent licensing of roxadustat for oral anemia therapy in China. Here, we survey current clinical experience with HIF-PHIs, discuss potential therapeutic advantages, and deliberate over safety concerns regarding long-term administration in patients with renal anemia.
Keywords: Anemia; Chronic kidney disease; Erythropoietin; Hypoxia-inducible factor; Prolyl hydroxylase domain.
Copyright © 2019 National Kidney Foundation, Inc. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures
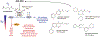
Similar articles
-
HIF-prolyl hydroxylases as therapeutic targets in erythropoiesis and iron metabolism.Hemodial Int. 2017 Jun;21 Suppl 1(Suppl 1):S110-S124. doi: 10.1111/hdi.12567. Hemodial Int. 2017. PMID: 28449418 Free PMC article. Review.
-
Hypoxia-Inducible Factor Prolyl Hydroxylase Inhibitors: A Potential New Treatment for Anemia in Patients With CKD.Am J Kidney Dis. 2017 Jun;69(6):815-826. doi: 10.1053/j.ajkd.2016.12.011. Epub 2017 Feb 24. Am J Kidney Dis. 2017. PMID: 28242135 Review.
-
A Novel Hypoxia-Inducible Factor-Prolyl Hydroxylase Inhibitor (GSK1278863) for Anemia in CKD: A 28-Day, Phase 2A Randomized Trial.Am J Kidney Dis. 2016 Jun;67(6):861-71. doi: 10.1053/j.ajkd.2015.11.021. Epub 2016 Jan 27. Am J Kidney Dis. 2016. PMID: 26827289 Clinical Trial.
-
Daprodustat: A Hypoxia-Inducible Factor-Prolyl Hydroxylase Inhibitor for Anemia of Chronic Kidney Disease.Ann Pharmacother. 2025 Jan;59(1):71-80. doi: 10.1177/10600280241241563. Epub 2024 Apr 14. Ann Pharmacother. 2025. PMID: 38616529 Review.
-
Nonclinical Characterization of the Hypoxia-Inducible Factor Prolyl Hydroxylase Inhibitor Roxadustat, a Novel Treatment of Anemia of Chronic Kidney Disease.J Pharmacol Exp Ther. 2020 Aug;374(2):342-353. doi: 10.1124/jpet.120.265181. Epub 2020 Jun 2. J Pharmacol Exp Ther. 2020. PMID: 32487538
Cited by
-
Erythropoietin regulation of red blood cell production: from bench to bedside and back.F1000Res. 2020 Sep 18;9:F1000 Faculty Rev-1153. doi: 10.12688/f1000research.26648.1. eCollection 2020. F1000Res. 2020. PMID: 32983414 Free PMC article. Review.
-
Roxadustat: Not just for anemia.Front Pharmacol. 2022 Aug 29;13:971795. doi: 10.3389/fphar.2022.971795. eCollection 2022. Front Pharmacol. 2022. PMID: 36105189 Free PMC article. Review.
-
Erythropoietin regulates metabolic response in mice via receptor expression in adipose tissue, brain, and bone.Exp Hematol. 2020 Dec;92:32-42. doi: 10.1016/j.exphem.2020.09.190. Epub 2020 Sep 18. Exp Hematol. 2020. PMID: 32950599 Free PMC article. Review.
-
Hypoxia-Induced circRNAs in Human Diseases: From Mechanisms to Potential Applications.Cells. 2022 Apr 19;11(9):1381. doi: 10.3390/cells11091381. Cells. 2022. PMID: 35563687 Free PMC article. Review.
-
Hypoxia inducible factors regulate hepatitis B virus replication by activating the basal core promoter.J Hepatol. 2021 Jul;75(1):64-73. doi: 10.1016/j.jhep.2020.12.034. Epub 2021 Jan 29. J Hepatol. 2021. PMID: 33516779 Free PMC article.
References
-
- Drueke TB, Locatelli F, Clyne N, et al. Normalization of hemoglobin level in patients with chronic kidney disease and anemia. N Engl J Med. 2006;355(20):2071–2084. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

