Surfactin and fengycin contribute to the protection of a Bacillus subtilis strain against grape downy mildew by both direct effect and defence stimulation
- PMID: 31104350
- PMCID: PMC6640177
- DOI: 10.1111/mpp.12809
Surfactin and fengycin contribute to the protection of a Bacillus subtilis strain against grape downy mildew by both direct effect and defence stimulation
Abstract
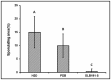
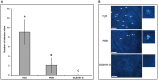
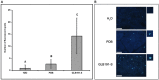
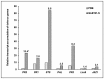
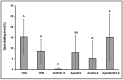
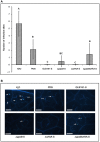
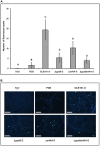
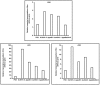
Bacillus subtilis GLB191 (hereafter GLB191) is an efficient biological control agent against the biotrophic oomycete Plasmopara viticola, the causal agent of grapevine downy mildew. In this study, we show that GLB191 supernatant is also highly active against downy mildew and that the activity results from both direct effect against the pathogen and stimulation of the plant defences (induction of defence gene expression and callose production). High-performance thin-layer chromatography analysis revealed the presence of the cyclic lipopeptides fengycin and surfactin in the supernatant. Mutants affected in the production of fengycin and/or surfactin were thus obtained and allowed us to show that both surfactin and fengycin contribute to the double activity of GLB191 supernatant against downy mildew. Altogether, this study suggests that GLB191 supernatant could be used as a new biocontrol product against grapevine downy mildew.
Keywords: Bacillus subtilis; Plasmopara viticola; defence; downy mildew; fengycin; lipopeptide; surfactin.
© 2019 The Authors. Molecular Plant Pathology published by British Society for Plant Pathology and John Wiley & Sons Ltd.
Figures
Similar articles
-
The Potential of Microorganisms for the Control of Grape Downy Mildew-A Review.J Fungi (Basel). 2024 Oct 8;10(10):702. doi: 10.3390/jof10100702. J Fungi (Basel). 2024. PMID: 39452654 Free PMC article. Review.
-
[Colonization of grape leaves by endophytic Bacillus subtilis JL4 and its control of grape downy mildew].Ying Yong Sheng Tai Xue Bao. 2016 Dec;27(12):4022-4028. doi: 10.13287/j.1001-9332.201612.038. Ying Yong Sheng Tai Xue Bao. 2016. PMID: 29704363 Chinese.
-
Proteomics towards the understanding of elicitor induced resistance of grapevine against downy mildew.J Proteomics. 2017 Mar 6;156:113-125. doi: 10.1016/j.jprot.2017.01.016. Epub 2017 Jan 31. J Proteomics. 2017. PMID: 28153682
-
Cyclic lipopeptides from Bacillus subtilis activate distinct patterns of defence responses in grapevine.Mol Plant Pathol. 2015 Feb;16(2):177-87. doi: 10.1111/mpp.12170. Epub 2014 Aug 24. Mol Plant Pathol. 2015. PMID: 25040001 Free PMC article.
-
Advances in understanding grapevine downy mildew: From pathogen infection to disease management.Mol Plant Pathol. 2024 Jan;25(1):e13401. doi: 10.1111/mpp.13401. Epub 2023 Nov 22. Mol Plant Pathol. 2024. PMID: 37991155 Free PMC article. Review.
Cited by
-
The Potential of Microorganisms for the Control of Grape Downy Mildew-A Review.J Fungi (Basel). 2024 Oct 8;10(10):702. doi: 10.3390/jof10100702. J Fungi (Basel). 2024. PMID: 39452654 Free PMC article. Review.
-
Genome sequence and comparative analysis of fungal antagonistic strain Bacillus velezensis LJBV19.Folia Microbiol (Praha). 2023 Feb;68(1):73-86. doi: 10.1007/s12223-022-00996-z. Epub 2022 Aug 1. Folia Microbiol (Praha). 2023. PMID: 35913660
-
Genomic insights into biocontrol potential of Bacillus stercoris LJBS06.3 Biotech. 2021 Nov;11(11):458. doi: 10.1007/s13205-021-03000-6. Epub 2021 Oct 9. 3 Biotech. 2021. PMID: 34692367 Free PMC article.
-
In Silico and In Vitro Evaluation of the Antimicrobial Potential of Bacillus cereus Isolated from Apis dorsata Gut against Neisseria gonorrhoeae.Antibiotics (Basel). 2021 Nov 15;10(11):1401. doi: 10.3390/antibiotics10111401. Antibiotics (Basel). 2021. PMID: 34827339 Free PMC article.
-
Exploiting RNA thermometer-driven molecular bioprocess control as a concept for heterologous rhamnolipid production.Sci Rep. 2021 Jul 20;11(1):14802. doi: 10.1038/s41598-021-94400-4. Sci Rep. 2021. PMID: 34285304 Free PMC article.
References
-
- Abdalla, M.A. , Win, H.Y. , Islam, M.T. , Von Tiedemann, A. , Schüffler, A. and Laatsch, H. (2011) Khatmiamycin, a motility inhibitor and zoosporicide against the grapevine downy mildew pathogen Plasmopara viticola from Streptomyces sp. ANK313. J. Antibiot. 64, 655. - PubMed
-
- Boutrot, F. and Zipfel, C. (2017) Function, discovery, and exploitation of plant pattern recognition receptors for broad‐spectrum disease resistance. Annu Rev Phytopathol. 55, 257–286. - PubMed
-
- Cawoy, H. , Mariutto, M. , Henry, G. , Fisher, C. , Vasilyeva, N. , Thonart, P. , Dommes, J. and Ongena, M. (2014) Plant defense stimulation by natural isolates of Bacillus depends on efficient surfactin production. Mol. Plant‐Microbe Interact. 27, 87–100. - PubMed
-
- Chandler, S. , Van Hese, N. , Coutte, F. , Jacques, P. , Höfte, M. and De Vleesschauwer, D. (2015) Role of cyclic lipopeptides produced by Bacillus subtilis in mounting induced immunity in rice (Oryza sativa L.). Physiol. Mol. Plant P. 91, 20–30.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

