EAP1 regulation of GnRH promoter activity is important for human pubertal timing
- PMID: 30608578
- PMCID: PMC6452208
- DOI: 10.1093/hmg/ddy451
EAP1 regulation of GnRH promoter activity is important for human pubertal timing
Abstract
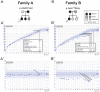
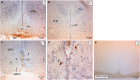
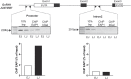
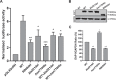
The initiation of puberty is orchestrated by an augmentation of gonadotropin-releasing hormone (GnRH) secretion from a few thousand hypothalamic neurons. Recent findings have indicated that the neuroendocrine control of puberty may be regulated by a hierarchically organized network of transcriptional factors acting upstream of GnRH. These include enhanced at puberty 1 (EAP1), which contributes to the initiation of female puberty through transactivation of the GnRH promoter. However, no EAP1 mutations have been found in humans with disorders of pubertal timing. We performed whole-exome sequencing in 67 probands and 93 relatives from a large cohort of familial self-limited delayed puberty (DP). Variants were analyzed for rare, potentially pathogenic variants enriched in case versus controls and relevant to the biological control of puberty. We identified one in-frame deletion (Ala221del) and one rare missense variant (Asn770His) in EAP1 in two unrelated families; these variants were highly conserved and potentially pathogenic. Expression studies revealed Eap1 mRNA abundance in peri-pubertal mouse hypothalamus. EAP1 binding to the GnRH1 promoter increased in monkey hypothalamus at the onset of puberty as determined by chromatin immunoprecipitation. Using a luciferase reporter assay, EAP1 mutants showed a reduced ability to trans-activate the GnRH promoter compared to wild-type EAP1, due to reduced protein levels caused by the Ala221del mutation and subcellular mislocation caused by the Asn770His mutation, as revealed by western blot and immunofluorescence, respectively. In conclusion, we have identified the first EAP1 mutations leading to reduced GnRH transcriptional activity resulting in a phenotype of self-limited DP.
© The Author(s) 2019. Published by Oxford University Press.
Figures

Similar articles
-
Genetics of pubertal timing.Best Pract Res Clin Endocrinol Metab. 2022 Jan;36(1):101618. doi: 10.1016/j.beem.2022.101618. Epub 2022 Feb 5. Best Pract Res Clin Endocrinol Metab. 2022. PMID: 35183440 Free PMC article. Review.
-
Transcription of the human EAP1 gene is regulated by upstream components of a puberty-controlling Tumor Suppressor Gene network.Mol Cell Endocrinol. 2012 Apr 4;351(2):184-98. doi: 10.1016/j.mce.2011.12.004. Epub 2011 Dec 19. Mol Cell Endocrinol. 2012. PMID: 22209758 Free PMC article.
-
Expression of EAP1 and CUX1 in the hypothalamus of female rats and relationship with KISS1 and GnRH.Endocr J. 2016 Aug 31;63(8):681-90. doi: 10.1507/endocrj.EJ16-0123. Epub 2016 May 31. Endocr J. 2016. PMID: 27250217
-
Enhanced at Puberty-1 (Eap1) Expression Critically Regulates the Onset of Puberty Independent of Hypothalamic Kiss1 Expression.Cell Physiol Biochem. 2017;43(4):1402-1412. doi: 10.1159/000481872. Epub 2017 Oct 11. Cell Physiol Biochem. 2017. PMID: 29017168
-
Genetics defects in GNRH1: a paradigm of hypothalamic congenital gonadotropin deficiency.Brain Res. 2010 Dec 10;1364:3-9. doi: 10.1016/j.brainres.2010.09.084. Epub 2010 Sep 29. Brain Res. 2010. PMID: 20887715 Review.
Cited by
-
Genetic, epigenetic and enviromental influencing factors on the regulation of precocious and delayed puberty.Front Endocrinol (Lausanne). 2022 Dec 22;13:1019468. doi: 10.3389/fendo.2022.1019468. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36619551 Free PMC article. Review.
-
Disorders of puberty and neurodevelopment: A shared etiology?Ann N Y Acad Sci. 2024 Nov;1541(1):83-99. doi: 10.1111/nyas.15246. Epub 2024 Oct 21. Ann N Y Acad Sci. 2024. PMID: 39431640 Free PMC article. Review.
-
Genetics of pubertal timing.Best Pract Res Clin Endocrinol Metab. 2022 Jan;36(1):101618. doi: 10.1016/j.beem.2022.101618. Epub 2022 Feb 5. Best Pract Res Clin Endocrinol Metab. 2022. PMID: 35183440 Free PMC article. Review.
-
Genetic architecture of self-limited delayed puberty and congenital hypogonadotropic hypogonadism.Front Endocrinol (Lausanne). 2023 Jan 16;13:1069741. doi: 10.3389/fendo.2022.1069741. eCollection 2022. Front Endocrinol (Lausanne). 2023. PMID: 36726466 Free PMC article. Review.
-
The Genetic Basis of Delayed Puberty.Front Endocrinol (Lausanne). 2019 Jun 26;10:423. doi: 10.3389/fendo.2019.00423. eCollection 2019. Front Endocrinol (Lausanne). 2019. PMID: 31293522 Free PMC article. Review.
References
-
- Kaprio J., Rimpelä A., Winter T., Viken R.J., Rimpelä M. and Rose R.J. (1995) Common genetic influences on BMI and age at menarche. Hum. Biol., 67, 739–753. - PubMed
-
- Juul A., Teilmann G., Scheike T., Hertel N.T., Holm K., Laursen E.M., Main K.M. and Skakkebaek N.E. (2006) Pubertal development in Danish children: comparison of recent European and US data. Int. J. Androl., 29, 247, 286–255; discussion, 290. - PubMed
-
- Teilmann G., Pedersen C.B., Skakkebaek N.E. and Jensen T.K. (2006) Increased risk of precocious puberty in internationally adopted children in Denmark. Pediatrics, 118, e391–e399. - PubMed
-
- Parent A.S., Teilmann G., Juul A., Skakkebaek N.E., Toppari J. and Bourguignon J.P. (2003) The timing of normal puberty and the age limits of sexual precocity: variations around the world, secular trends, and changes after migration. Endocr. Rev., 24, 668–693. - PubMed
-
- Morris D.H., Jones M.E., Schoemaker M.J., Ashworth A. and Swerdlow A.J. (2011) Familial concordance for age at menarche: analyses from the Breakthrough Generations Study. Paediatr. Perinat. Epidemiol., 25, 306–311. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

