Intermittent androgen deprivation therapy: recommendations to improve the management of patients with prostate cancer following the GRADE approach
- PMID: 30122985
- PMCID: PMC6080876
- DOI: 10.2147/CMAR.S164856
Intermittent androgen deprivation therapy: recommendations to improve the management of patients with prostate cancer following the GRADE approach
Abstract
Purpose: The purpose of this study was to provide evidence-based recommendations of intermittent androgen deprivation therapy (IADT) compared with continuous androgen deprivation therapy (CADT) for men with prostate cancer (PCA).
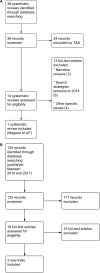
Methods: We conducted a comprehensive search in MEDLINE, EMBASE, The Cochrane Library, CINAHL, and ECONLIT, from the database inception to December 2017. We adhered to the Grading of Recommendations, Assessment, Development and Evaluation framework to assess the quality of the evidence and to formulate recommendations.
Results: We included one systematic review with 15 trials as well as three additional studies that assessed IADT versus CADT, all of them focused on PCA patients in advanced stages. The findings did not show differences for critical and important outcomes, including adverse events. Trials reported the benefits of IADT in terms of selected domains of health-related quality of life, although with high heterogeneity. Evidence quality was considered moderate or low for most of the assessed outcomes. We identified a patient preference study reporting a high preference for IADT, due to issues related to quality of life, general well-being, and side effects, among others. We did not identify economic studies comparing these regimes. We formulate four recommendations: one no-recommendation, one conditional recommendation, and two good practice points.
Conclusion: For men in early stages of PCA, it is not possible to make any recommendation about the preferable use of IADT or CADT due to the lack of available evidence. For men in advanced stages of the disease, an IADT should be considered as soon as clinically reasonable (weak recommendation and low certainty of the evidence). Clinicians should discuss the risks and benefits of IADT and CADT with their patients, taking into account their values and preferences.
Keywords: GRADE approach; evidence-based medicine; hormone deprivation therapy; prostate cancer; prostate neoplasm.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures
Similar articles
-
[Risks of diabetes mellitus and impaired glucose tolerance induced by intermittent versus continuous androgen-deprivation therapy for advanced prostate cancer].Zhonghua Nan Ke Xue. 2017 Jul;23(7):598-602. Zhonghua Nan Ke Xue. 2017. PMID: 29723451 Chinese.
-
Efficacy of intermittent androgen deprivation therapy vs conventional continuous androgen deprivation therapy for advanced prostate cancer: a meta-analysis.Urology. 2013 Aug;82(2):327-33. doi: 10.1016/j.urology.2013.01.078. Urology. 2013. PMID: 23896094 Free PMC article.
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
A meta-analysis of cardiovascular events in intermittent androgen-deprivation therapy versus continuous androgen-deprivation therapy for prostate cancer patients.Prostate Cancer Prostatic Dis. 2016 Dec;19(4):333-339. doi: 10.1038/pcan.2016.35. Epub 2016 Sep 6. Prostate Cancer Prostatic Dis. 2016. PMID: 27595915 Review.
-
Intermittent androgen deprivation therapy for prostate cancer: translating randomized controlled trials into clinical practice.Can J Urol. 2014 Apr;21(2 Supp 1):28-36. Can J Urol. 2014. PMID: 24775721 Review.
Cited by
-
Cabazitaxel suppresses the proliferation and promotes the apoptosis and radiosensitivity of castration-resistant prostate cancer cells by inhibiting PI3K/AKT pathway.Am J Transl Res. 2022 Jan 15;14(1):166-181. eCollection 2022. Am J Transl Res. 2022. PMID: 35173836 Free PMC article.
-
Management algorithms for metastatic prostate cancer.Can Urol Assoc J. 2020 Feb;14(2):50-60. doi: 10.5489/cuaj.5840. Can Urol Assoc J. 2020. PMID: 31039111 Free PMC article. Review.
-
Guidance for the assessment and management of prostate cancer treatment-induced bone loss. A consensus position statement from an expert group.J Bone Oncol. 2020 Aug 2;25:100311. doi: 10.1016/j.jbo.2020.100311. eCollection 2020 Dec. J Bone Oncol. 2020. PMID: 32995252 Free PMC article.
-
Treatment-Related Cognitive Impairment in Patients with Prostate Cancer: Patients' Real-World Insights for Optimizing Outcomes.Adv Ther. 2024 Feb;41(2):476-491. doi: 10.1007/s12325-023-02721-9. Epub 2023 Nov 18. Adv Ther. 2024. PMID: 37979089 Free PMC article. Review.
-
Intermittent versus continuous androgen deprivation therapy for advanced prostate cancer.Nat Rev Urol. 2020 Aug;17(8):469-481. doi: 10.1038/s41585-020-0335-7. Epub 2020 Jun 30. Nat Rev Urol. 2020. PMID: 32606361 Review.
References
-
- Carroll PR, Parsons JK, Andriole G, et al. NCCN Clinical Practice Guidelines Prostate Cancer Early Detection, Version 2.2015. J Natl Compr Canc Netw. 2015;13(12):1534–1561. - PubMed
-
- Giacalone A, Quitadamo D, Zanet E, Berretta M, Spina M, Tirelli U. Cancer-related fatigue in the elderly. Support Care Cancer. 2013;21(10):2899–2911. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources