Outcomes of patients with myelofibrosis treated with compassionate use pacritinib: a sponsor-independent international study
- PMID: 29616317
- PMCID: PMC6019145
- DOI: 10.1007/s00277-018-3309-6
Outcomes of patients with myelofibrosis treated with compassionate use pacritinib: a sponsor-independent international study
Abstract
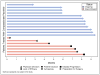
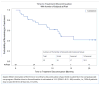
Myelofibrosis (MF) is a chronic yet progressive myeloid neoplasm in which only a minority of patients undergo curative therapy, hematopoietic stem cell transplantation. Ruxolitinib, a JAK1/2 inhibitor, is the lone therapy approved for MF, offering a clear symptom and spleen benefit at the expense of treatment-related cytopenias. Pacritinib (PAC), a multi-kinase inhibitor with specificity for JAK2, FLT3, and IRAK1 but sparing JAK1, has demonstrated clinical activity in MF with minimal myelosuppression. Due to an FDA-mandated full clinical hold, the randomized phase 3 PERSIST trials were abruptly stopped and PAC was immediately discontinued for all patients. Thirty-three patients benefitting from PAC on clinical trial prior to the hold were allowed to resume therapy on an individual, compassionate-use basis. This study reports the detailed outcomes of 19 of these PAC retreatment patients with a median follow-up of 8 months. Despite a median platelet count of 49 × 109/L at restart of PAC, no significant change in hematologic profile was observed. Grade 3/4 adverse events of epistaxis (n = 1), asymptomatic QT prolongation (n = 1), and bradycardia (n = 1) occurred in three patients within the first 3 months of retreatment. One death due to catheter-associated sepsis occurred. The median time to discontinuation of PAC therapy on compassionate use for all 33 patients was 12.2 (95% CI 8.3-NR) months. PAC retreatment was associated with modest improvement in splenomegaly without progressive myelosuppression and supports the continued development of this agent for the treatment of MF second line to ruxolitinib or in the setting of treatment-limiting thrombocytopenia.
Keywords: JAK inhibitor; Myelofibrosis; Pacritinib.
Conflict of interest statement
Conflicts of Interest: The authors declare no conflicts of interest.
Figures
Similar articles
-
Pacritinib vs Best Available Therapy, Including Ruxolitinib, in Patients With Myelofibrosis: A Randomized Clinical Trial.JAMA Oncol. 2018 May 1;4(5):652-659. doi: 10.1001/jamaoncol.2017.5818. JAMA Oncol. 2018. PMID: 29522138 Free PMC article. Clinical Trial.
-
Pacritinib to treat myelofibrosis patients with thrombocytopenia.Expert Rev Hematol. 2018 Sep;11(9):707-714. doi: 10.1080/17474086.2018.1500456. Epub 2018 Jul 19. Expert Rev Hematol. 2018. PMID: 30001163 Review.
-
Pacritinib versus best available therapy for the treatment of myelofibrosis irrespective of baseline cytopenias (PERSIST-1): an international, randomised, phase 3 trial.Lancet Haematol. 2017 May;4(5):e225-e236. doi: 10.1016/S2352-3026(17)30027-3. Epub 2017 Mar 20. Lancet Haematol. 2017. PMID: 28336242 Free PMC article. Clinical Trial.
-
Phase 1/2 study of pacritinib, a next generation JAK2/FLT3 inhibitor, in myelofibrosis or other myeloid malignancies.J Hematol Oncol. 2016 Dec 8;9(1):137. doi: 10.1186/s13045-016-0367-x. J Hematol Oncol. 2016. PMID: 27931243 Free PMC article. Clinical Trial.
-
A comprehensive review of pacritinib in myelofibrosis.Future Oncol. 2015;11(20):2819-30. doi: 10.2217/fon.15.200. Epub 2015 Sep 14. Future Oncol. 2015. PMID: 26367195 Free PMC article. Review.
Cited by
-
Classical Philadelphia-negative myeloproliferative neoplasms: focus on mutations and JAK2 inhibitors.Med Oncol. 2018 Aug 3;35(9):119. doi: 10.1007/s12032-018-1187-3. Med Oncol. 2018. PMID: 30074114 Free PMC article. Review.
-
Beyond Ruxolitinib: Fedratinib and Other Emergent Treatment Options for Myelofibrosis.Cancer Manag Res. 2019 Dec 24;11:10777-10790. doi: 10.2147/CMAR.S212559. eCollection 2019. Cancer Manag Res. 2019. PMID: 31920387 Free PMC article. Review.
-
Molecular Genetics of Thrombotic Myeloproliferative Neoplasms: Implications in Precision Oncology.Diagnostics (Basel). 2023 Jan 3;13(1):163. doi: 10.3390/diagnostics13010163. Diagnostics (Basel). 2023. PMID: 36611455 Free PMC article. Review.
-
The application of JAK inhibitors in the peri-transplantation period of hematopoietic stem cell transplantation for myelofibrosis.Ann Hematol. 2024 Sep;103(9):3293-3301. doi: 10.1007/s00277-024-05703-1. Epub 2024 Mar 18. Ann Hematol. 2024. PMID: 38494551 Free PMC article. Review.
References
-
- Hoffman R, Rondelli D. Biology and treatment of primary myelofibrosis. Hematology Am Soc Hematol Educ Program. 2007:346–54. - PubMed
-
- Mascarenhas JO, Cross NC, Mesa RA. The future of JAK inhibition in myelofibrosis and beyond. Blood Rev. 2014 Sep;28(5):189–96. - PubMed
-
- Harrison C, Kiladjian JJ, Al-Ali HK, Gisslinger H, Waltzman R, Stalbovskaya V, et al. JAK inhibition with ruxolitinib versus best available therapy for myelofibrosis. N Engl J Med. 2012 Mar 1;366(9):787–98. - PubMed
-
- Cervantes F, Pereira A. Does ruxolitinib prolong the survival of patients with myelofibrosis? Blood. 2017 Feb 16;129(7):832–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

