Association Between Amyloid and Tau Accumulation in Young Adults With Autosomal Dominant Alzheimer Disease
- PMID: 29435558
- PMCID: PMC5885174
- DOI: 10.1001/jamaneurol.2017.4907
Association Between Amyloid and Tau Accumulation in Young Adults With Autosomal Dominant Alzheimer Disease
Abstract
Importance: It is critically important to improve our ability to diagnose and track Alzheimer disease (AD) as early as possible. Individuals with autosomal dominant forms of AD can provide clues as to which and when biological changes are reliably present prior to the onset of clinical symptoms.
Objective: To characterize the associations between amyloid and tau deposits in the brains of cognitively unimpaired and impaired carriers of presenilin 1 (PSEN1) E280A mutation.
Design, setting, and participants: In this cross-sectional imaging study, we leveraged data from a homogeneous autosomal dominant AD kindred, which allowed us to examine measurable tau deposition as a function of individuals' proximity to the expected onset of dementia. Cross-sectional measures of carbon 11-labeled Pittsburgh Compound B positron emission tomography (PET) and flortaucipir F 18 (previously known as AV 1451, T807) PET imaging were assessed in 24 PSEN1 E280A kindred members (age range, 28-55 years), including 12 carriers, 9 of whom were cognitively unimpaired and 3 of whom had mild cognitive impairment, and 12 cognitively unimpaired noncarriers.
Main outcomes and measures: We compared carbon 11-labeled Pittsburgh Compound B PET cerebral with cerebellar distribution volume ratios as well as flortaucipir F 18 PET cerebral with cerebellar standardized uptake value ratios in mutation carriers and noncarriers. Spearman correlations characterized the associations between age and mean cortical Pittsburgh Compound B distribution volume ratio levels or regional flortaucipir standardized uptake value ratio levels in both groups.
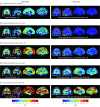
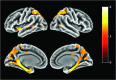
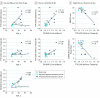
Results: Of the 24 individuals, the mean (SD) age was 38.0 (7.4) years, or approximately 6 years younger than the expected onset of clinical symptoms in carriers. Compared with noncarriers, cognitively unimpaired mutation carriers had elevated mean cortical Pittsburgh Compound B distribution volume ratio levels in their late 20s, and 7 of 9 carriers older than 30 years reached the threshold for amyloidosis (distribution volume ratio level > 1.2). Elevated levels of tau deposition were seen within medial temporal lobe regions in amyloid-positive mutation carriers 6 years before clinical onset of AD in this kindred. Substantial tau deposition in the neocortex was only observed in 1 unimpaired carrier and in those with mild cognitive impairment. β-Amyloid uptake levels were diffusely elevated in unimpaired carriers approximately 15 years prior to expected onset of mild cognitive impairment. In carriers, higher levels of tau deposition were associated with worse performance on the Mini-Mental State Examination (entorhinal cortex: r = -0.60; P = .04; inferior temporal lobe: r = -0.54; P = .06) and the Consortium to Establish a Registry for Alzheimer Disease Word List Delayed Recall (entorhinal cortex: r = -0.86; P < .001; inferior temporal lobe: r = -0.70; P = .01).
Conclusions and relevance: The present findings add to the growing evidence that molecular markers can characterize biological changes associated with AD in individuals who are still cognitively unimpaired. The findings also suggest that tau PET imaging may be useful as a biomarker to distinguish individuals at high risk to develop the clinical symptoms of AD and to track disease progression.
Conflict of interest statement
Figures
Dataset use reported in
-
Tau Positron Emission Tomography in Autosomal Dominant Alzheimer Disease: Small Windows, Big Picture.JAMA Neurol. 2018 May 1;75(5):536-538. doi: 10.1001/jamaneurol.2017.4026. JAMA Neurol. 2018. PMID: 29435570 Free PMC article. No abstract available.
Similar articles
-
Associations between biomarkers and age in the presenilin 1 E280A autosomal dominant Alzheimer disease kindred: a cross-sectional study.JAMA Neurol. 2015 Mar;72(3):316-24. doi: 10.1001/jamaneurol.2014.3314. JAMA Neurol. 2015. PMID: 25580592 Free PMC article.
-
Characterizing brain tau and cognitive decline along the amyloid timeline in Alzheimer's disease.Brain. 2024 Jun 3;147(6):2144-2157. doi: 10.1093/brain/awae116. Brain. 2024. PMID: 38667631
-
Comorbidities in Early-Onset Sporadic versus Presenilin-1 Mutation-Associated Alzheimer's Disease Dementia: Evidence for Dependency on Alzheimer's Disease Neuropathological Changes.medRxiv [Preprint]. 2023 Aug 16:2023.08.14.23294081. doi: 10.1101/2023.08.14.23294081. medRxiv. 2023. Update in: J Neuropathol Exp Neurol. 2025 Feb 1;84(2):104-113. doi: 10.1093/jnen/nlae122. PMID: 37646002 Free PMC article. Updated. Preprint.
-
The Alzheimer's prevention initiative composite cognitive test score: sample size estimates for the evaluation of preclinical Alzheimer's disease treatments in presenilin 1 E280A mutation carriers.J Clin Psychiatry. 2014 Jun;75(6):652-60. doi: 10.4088/JCP.13m08927. J Clin Psychiatry. 2014. PMID: 24816373 Free PMC article. Review.
-
Depressing time: Waiting, melancholia, and the psychoanalytic practice of care.In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. PMID: 36137063 Free Books & Documents. Review.
Cited by
-
The neural economics of brain aging.Sci Rep. 2021 Jun 9;11(1):12167. doi: 10.1038/s41598-021-91621-5. Sci Rep. 2021. PMID: 34108560 Free PMC article.
-
Spatial Relationships between Molecular Pathology and Neurodegeneration in the Alzheimer's Disease Continuum.Cereb Cortex. 2021 Jan 1;31(1):1-14. doi: 10.1093/cercor/bhaa184. Cereb Cortex. 2021. PMID: 32808011 Free PMC article.
-
Dissecting the clinical heterogeneity of early-onset Alzheimer's disease.Mol Psychiatry. 2022 Jun;27(6):2674-2688. doi: 10.1038/s41380-022-01531-9. Epub 2022 Apr 7. Mol Psychiatry. 2022. PMID: 35393555 Free PMC article. Review.
-
Biomarkers for tau pathology.Mol Cell Neurosci. 2019 Jun;97:18-33. doi: 10.1016/j.mcn.2018.12.001. Epub 2018 Dec 7. Mol Cell Neurosci. 2019. PMID: 30529601 Free PMC article. Review.
-
Imaging the evolution and pathophysiology of Alzheimer disease.Nat Rev Neurosci. 2018 Nov;19(11):687-700. doi: 10.1038/s41583-018-0067-3. Nat Rev Neurosci. 2018. PMID: 30266970 Free PMC article. Review.
References
-
- Iqbal K, Grundke-Iqbal I. Neurofibrillary pathology leads to synaptic loss and not the other way around in Alzheimer disease. J Alzheimers Dis. 2002;4(3):235-238. - PubMed
-
- Arnold SE, Hyman BT, Flory J, Damasio AR, Van Hoesen GW. The topographical and neuroanatomical distribution of neurofibrillary tangles and neuritic plaques in the cerebral cortex of patients with Alzheimer’s disease. Cereb Cortex. 1991;1(1):103-116. - PubMed
-
- Braak H, Braak E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991;82(4):239-259. - PubMed
-
- Lopera F, Ardilla A, Martínez A, et al. . Clinical features of early-onset Alzheimer disease in a large kindred with an E280A presenilin-1 mutation. JAMA. 1997;277(10):793-799. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

