Tissue Distribution and Elimination of Isavuconazole following Single and Repeat Oral-Dose Administration of Isavuconazonium Sulfate to Rats
- PMID: 28971866
- PMCID: PMC5700325
- DOI: 10.1128/AAC.01292-17
Tissue Distribution and Elimination of Isavuconazole following Single and Repeat Oral-Dose Administration of Isavuconazonium Sulfate to Rats
Abstract
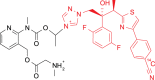
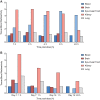
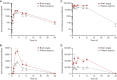
Quantitative whole-body autoradiography was used to assess the distribution and tissue penetration of isavuconazole in rats following single and repeated oral-dose administration of radiolabeled isavuconazonium sulfate, the prodrug of isavuconazole. Following a single-dose administration of radiolabeled isavuconazonium sulfate (labeled on the active moiety), radioactivity was detectable within 1 h postdose in 56 of 65 tissue/fluid specimens. The highest maximum concentrations (Cmax) were observed in bile and liver (66.6 and 24.7 μg eq/g, respectively). The lowest Cmax values were in bone and eye lens (0.070 and 0.077 μg eq/g, respectively). By 144 h postdose, radioactivity was undetectable in all tissues/fluids except liver (undetectable at 336 h) and adrenal gland tissues (undetectable at 672 h). Following daily administration for up to 21 days, 1-h-postdose Cmax values were the highest on or before day 14 in all except seven tissues/fluids, of which only rectum mucosa and small intestine mucosa had Cmax values >25% higher than all other 1-h-postdose values. For 24-h-postdose Cmax values, only large intestine, large intestine mucosa, and urine had the highest Cmax values at day 21. The penetration of single oral doses of unlabeled isavuconazole (25 mg/kg of body weight isavuconazonium sulfate) and voriconazole (50 mg/kg) into rat brain (assessed using liquid chromatography-tandem mass spectrometry) was also compared. Brain concentration/plasma concentration ratios reached approximately 1.8:1 and 2:1, respectively. These data suggest that isavuconazole penetrates most tissues rapidly, reaches a steady state in most or all tissues/fluids within 14 days, does not accumulate in tissues/fluids over time, and achieves potentially efficacious concentrations in the brain.
Keywords: isavuconazole; isavuconazonium sulfate; quantitative whole-body autoradiography; rat; tissue distribution; tissue penetration.
Copyright © 2017 Schmitt-Hoffmann et al.
Figures
Similar articles
-
Tissue Distribution and Penetration of Isavuconazole at the Site of Infection in Experimental Invasive Aspergillosis in Mice with Underlying Chronic Granulomatous Disease.Antimicrob Agents Chemother. 2019 May 24;63(6):e00524-19. doi: 10.1128/AAC.00524-19. Print 2019 Jun. Antimicrob Agents Chemother. 2019. PMID: 30988140 Free PMC article.
-
Two Phase 1, Open-Label, Mass Balance Studies to Determine the Pharmacokinetics of 14 C-Labeled Isavuconazonium Sulfate in Healthy Male Volunteers.Clin Pharmacol Drug Dev. 2018 Feb;7(2):207-216. doi: 10.1002/cpdd.376. Epub 2017 Jul 27. Clin Pharmacol Drug Dev. 2018. PMID: 28750160 Free PMC article. Clinical Trial.
-
Isavuconazole absorption following oral administration in healthy subjects is comparable to intravenous dosing, and is not affected by food, or drugs that alter stomach pH.Int J Clin Pharmacol Ther. 2016 Aug;54(8):572-80. doi: 10.5414/CP202434. Int J Clin Pharmacol Ther. 2016. PMID: 27345284 Clinical Trial.
-
Isavuconazole: A new broad-spectrum azole. Part 2: pharmacokinetics and clinical activity.J Mycol Med. 2018 Mar;28(1):15-22. doi: 10.1016/j.mycmed.2018.02.002. Epub 2018 Mar 16. J Mycol Med. 2018. PMID: 29551442 Review.
-
Isavuconazonium sulfate: a triazole prodrug for invasive fungal infections.Int J Pharm Pract. 2017 Feb;25(1):18-30. doi: 10.1111/ijpp.12302. Epub 2016 Aug 29. Int J Pharm Pract. 2017. PMID: 27569742 Review.
Cited by
-
Approaches for Targeting Naegleria fowleri Using Nanoparticles and Artificial Peptides.Pathogens. 2024 Aug 16;13(8):695. doi: 10.3390/pathogens13080695. Pathogens. 2024. PMID: 39204295 Free PMC article. Review.
-
Case report: Successful combination therapy with isavuconazole and amphotericin B in treatment of disseminated Candida tropicalis infection.Front Med (Lausanne). 2024 Jun 24;11:1397539. doi: 10.3389/fmed.2024.1397539. eCollection 2024. Front Med (Lausanne). 2024. PMID: 38978781 Free PMC article.
-
Isavuconazole for Treatment of Experimental Fungal Endophthalmitis Caused by Aspergillus fumigatus.Antimicrob Agents Chemother. 2018 Oct 24;62(11):e01537-18. doi: 10.1128/AAC.01537-18. Print 2018 Nov. Antimicrob Agents Chemother. 2018. PMID: 30201814 Free PMC article.
-
Pharmacodynamics of Isavuconazole in a Rabbit Model of Cryptococcal Meningoencephalitis.Antimicrob Agents Chemother. 2019 Aug 23;63(9):e00546-19. doi: 10.1128/AAC.00546-19. Print 2019 Sep. Antimicrob Agents Chemother. 2019. PMID: 31209006 Free PMC article.
-
Beyond tissue concentrations: antifungal penetration at the site of infection.Med Mycol. 2019 Apr 1;57(Supplement_2):S161-S167. doi: 10.1093/mmy/myy067. Med Mycol. 2019. PMID: 30816968 Free PMC article. Review.
References
-
- Kontoyiannis DP, Marr KA, Park BJ, Alexander BD, Anaissie EJ, Walsh TJ, Ito J, Andes DR, Baddley JW, Brown JM, Brumble LM, Freifeld AG, Hadley S, Herwaldt LA, Kauffman CA, Knapp K, Lyon GM, Morrison VA, Papanicolaou G, Patterson TF, Perl TM, Schuster MG, Walker R, Wannemuehler KA, Wingard JR, Chiller TM, Pappas PG. 2010. Prospective surveillance for invasive fungal infections in hematopoietic stem cell transplant recipients, 2001-2006: overview of the Transplant-Associated Infection Surveillance Network (TRANSNET) database. Clin Infect Dis 50:1091–1100. doi:10.1086/651263. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

