Bacillomycin D Produced by Bacillus amyloliquefaciens Is Involved in the Antagonistic Interaction with the Plant-Pathogenic Fungus Fusarium graminearum
- PMID: 28733288
- PMCID: PMC5601353
- DOI: 10.1128/AEM.01075-17
Bacillomycin D Produced by Bacillus amyloliquefaciens Is Involved in the Antagonistic Interaction with the Plant-Pathogenic Fungus Fusarium graminearum
Abstract
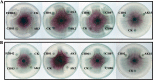
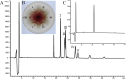
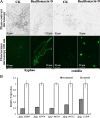
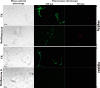
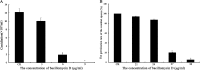
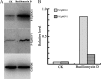
Fusarium graminearum (teleomorph: Ascomycota, Hypocreales, Gibberella, Gibberella zeae) is a destructive fungal pathogen that threatens the production and quality of wheat and barley worldwide. Controlling this toxin-producing pathogen is a significant challenge. In the present study, the commercially available strain Bacillus amyloliquefaciens (Bacteria, Firmicutes, Bacillales, Bacillus) FZB42 showed strong activity against F. graminearum The lipopeptide bacillomycin D, produced by FZB42, was shown to contribute to the antifungal activity. Purified bacillomycin D showed strong activity against F. graminearum, and its 50% effective concentration was determined to be approximately 30 μg/ml. Analyses using scanning and transmission electron microscopy revealed that bacillomycin D caused morphological changes in the plasma membranes and cell walls of F. graminearum hyphae and conidia. Fluorescence microscopy combined with different dyes showed that bacillomycin D induced the accumulation of reactive oxygen species and caused cell death in F. graminearum hyphae and conidia. F. graminearum secondary metabolism also responded to bacillomycin D challenge, by increasing the production of deoxynivalenol. Biological control experiments demonstrated that bacillomycin D exerted good control of F. graminearum on corn silks, wheat seedlings, and wheat heads. In response to bacillomycin D, F. graminearum genes involved in scavenging reactive oxygen species were downregulated, whereas genes involved in the synthesis of deoxynivalenol were upregulated. Phosphorylation of MGV1 and HOG1, the mitogen-activated protein kinases of F. graminearum, was increased in response to bacillomycin D. Taken together, these findings reveal the mechanism of the antifungal action of bacillomycin D.IMPORTANCE Biological control of plant disease caused by Fusarium graminearum is desirable. Bacillus amyloliquefaciens FZB42 is a representative of the biocontrol bacterial strains. In this work, the lipopeptide bacillomycin D, produced by FZB42, showed strong fungicidal activity against F. graminearum Bacillomycin D caused morphological changes in the plasma membrane and cell wall of F. graminearum, induced accumulation of reactive oxygen species, and ultimately caused cell death in F. graminearum Interestingly, when F. graminearum was challenged with bacillomycin D, the deoxynivalenol production, gene expression, mitogen-activated protein kinase phosphorylation, and pathogenicity of F. graminearum were significantly altered. These findings clarified the mechanisms of the activity of bacillomycin D against F. graminearum and highlighted the potential of B. amyloliquefaciens FZB42 as a biocontrol agent against F. graminearum.
Keywords: Bacillus amyloliquefaciens; Fusarium graminearum; bacillomycin D; cell death; fungus-bacterium interactions; mitogen-activated protein kinases; reactive oxygen species.
Copyright © 2017 American Society for Microbiology.
Figures



Similar articles
-
Fengycin Produced by Bacillus amyloliquefaciens FZB42 Inhibits Fusarium graminearum Growth and Mycotoxins Biosynthesis.Toxins (Basel). 2019 May 24;11(5):295. doi: 10.3390/toxins11050295. Toxins (Basel). 2019. PMID: 31137632 Free PMC article.
-
Antifungal efficacy of Bacillus amyloliquefaciens ZK-9 against Fusarium graminearum and analysis of the potential mechanism of its lipopeptides.Int J Food Microbiol. 2024 Sep 16;422:110821. doi: 10.1016/j.ijfoodmicro.2024.110821. Epub 2024 Jul 1. Int J Food Microbiol. 2024. PMID: 38970998
-
Antifungal potential of lipopeptides produced by the Bacillus siamensis Sh420 strain against Fusarium graminearum.Microbiol Spectr. 2024 Apr 2;12(4):e0400823. doi: 10.1128/spectrum.04008-23. Epub 2024 Mar 7. Microbiol Spectr. 2024. PMID: 38451229 Free PMC article.
-
Surfactin inhibits Fusarium graminearum by accumulating intracellular ROS and inducing apoptosis mechanisms.World J Microbiol Biotechnol. 2023 Oct 12;39(12):340. doi: 10.1007/s11274-023-03790-2. World J Microbiol Biotechnol. 2023. PMID: 37821760 Review.
-
Recent advances in genes involved in secondary metabolite synthesis, hyphal development, energy metabolism and pathogenicity in Fusarium graminearum (teleomorph Gibberella zeae).Biotechnol Adv. 2014 Mar-Apr;32(2):390-402. doi: 10.1016/j.biotechadv.2013.12.007. Epub 2014 Jan 2. Biotechnol Adv. 2014. PMID: 24389085 Review.
Cited by
-
Iturin A Extracted From Bacillus subtilis WL-2 Affects Phytophthora infestans via Cell Structure Disruption, Oxidative Stress, and Energy Supply Dysfunction.Front Microbiol. 2020 Sep 9;11:536083. doi: 10.3389/fmicb.2020.536083. eCollection 2020. Front Microbiol. 2020. PMID: 33013776 Free PMC article.
-
Jeongeuplla avenae gen. nov., sp. nov., a novel β-carotene-producing bacterium that alleviates salinity stress in Arabidopsis.Front Microbiol. 2023 Dec 6;14:1265308. doi: 10.3389/fmicb.2023.1265308. eCollection 2023. Front Microbiol. 2023. PMID: 38125566 Free PMC article.
-
Soil Bacteria Isolated From Tunisian Arid Areas Show Promising Antimicrobial Activities Against Gram-Negatives.Front Microbiol. 2018 Nov 13;9:2742. doi: 10.3389/fmicb.2018.02742. eCollection 2018. Front Microbiol. 2018. PMID: 30483240 Free PMC article.
-
Efficacy of biological agents and compost on growth and resistance of tomatoes to late blight.Planta. 2019 Mar;249(3):799-813. doi: 10.1007/s00425-018-3035-2. Epub 2018 Nov 7. Planta. 2019. PMID: 30406411
-
Characterization of Bacillus velezensis UTB96, Demonstrating Improved Lipopeptide Production Compared to the Strain B. velezensis FZB42.Microorganisms. 2022 Nov 10;10(11):2225. doi: 10.3390/microorganisms10112225. Microorganisms. 2022. PMID: 36363818 Free PMC article.
References
-
- Mesterhazy A. 1995. Types and components of resistance to Fusarium head blight. Plant Breed 114:377–386. doi:10.1111/j.1439-0523.1995.tb00816.x. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

