Systematic review adherence to methodological or reporting quality
- PMID: 28720117
- PMCID: PMC5516390
- DOI: 10.1186/s13643-017-0527-2
Systematic review adherence to methodological or reporting quality
Abstract
Background: Guidelines for assessing methodological and reporting quality of systematic reviews (SRs) were developed to contribute to implementing evidence-based health care and the reduction of research waste. As SRs assessing a cohort of SRs is becoming more prevalent in the literature and with the increased uptake of SR evidence for decision-making, methodological quality and standard of reporting of SRs is of interest. The objective of this study is to evaluate SR adherence to the Quality of Reporting of Meta-analyses (QUOROM) and PRISMA reporting guidelines and the A Measurement Tool to Assess Systematic Reviews (AMSTAR) and Overview Quality Assessment Questionnaire (OQAQ) quality assessment tools as evaluated in methodological overviews.
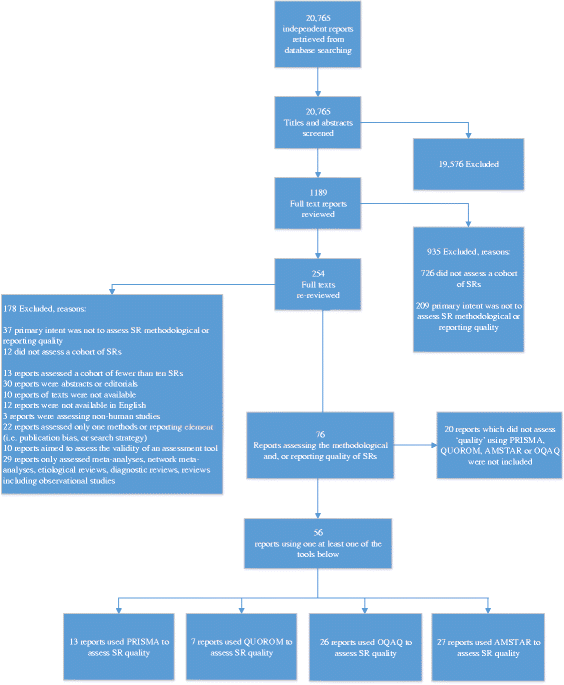
Methods: The Cochrane Library, MEDLINE®, and EMBASE® databases were searched from January 1990 to October 2014. Title and abstract screening and full-text screening were conducted independently by two reviewers. Reports assessing the quality or reporting of a cohort of SRs of interventions using PRISMA, QUOROM, OQAQ, or AMSTAR were included. All results are reported as frequencies and percentages of reports and SRs respectively.
Results: Of the 20,765 independent records retrieved from electronic searching, 1189 reports were reviewed for eligibility at full text, of which 56 reports (5371 SRs in total) evaluating the PRISMA, QUOROM, AMSTAR, and/or OQAQ tools were included. Notable items include the following: of the SRs using PRISMA, over 85% (1532/1741) provided a rationale for the review and less than 6% (102/1741) provided protocol information. For reports using QUOROM, only 9% (40/449) of SRs provided a trial flow diagram. However, 90% (402/449) described the explicit clinical problem and review rationale in the introduction section. Of reports using AMSTAR, 30% (534/1794) used duplicate study selection and data extraction. Conversely, 80% (1439/1794) of SRs provided study characteristics of included studies. In terms of OQAQ, 37% (499/1367) of the SRs assessed risk of bias (validity) in the included studies, while 80% (1112/1387) reported the criteria for study selection.
Conclusions: Although reporting guidelines and quality assessment tools exist, reporting and methodological quality of SRs are inconsistent. Mechanisms to improve adherence to established reporting guidelines and methodological assessment tools are needed to improve the quality of SRs.
Keywords: Guideline adherence; Methodological quality; Reporting quality; Systematic reviews.
Similar articles
-
Identifying approaches for assessing methodological and reporting quality of systematic reviews: a descriptive study.Syst Rev. 2017 Jun 19;6(1):117. doi: 10.1186/s13643-017-0507-6. Syst Rev. 2017. PMID: 28629396 Free PMC article. Review.
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
Methodological and reporting quality assessment of systematic reviews and meta-analyses in the association between sleep duration and hypertension.Syst Rev. 2024 Aug 6;13(1):211. doi: 10.1186/s13643-024-02622-0. Syst Rev. 2024. PMID: 39107813 Free PMC article.
-
Abstract analysis method facilitates filtering low-methodological quality and high-bias risk systematic reviews on psoriasis interventions.BMC Med Res Methodol. 2017 Dec 29;17(1):180. doi: 10.1186/s12874-017-0460-z. BMC Med Res Methodol. 2017. PMID: 29284417 Free PMC article.
-
Poor methodological quality and reporting standards of systematic reviews in burn care management.Int Wound J. 2017 Oct;14(5):754-763. doi: 10.1111/iwj.12692. Epub 2016 Dec 18. Int Wound J. 2017. PMID: 27990772 Free PMC article. Review.
Cited by
-
Effect of PRISMA 2009 on reporting quality in systematic reviews and meta-analyses in high-impact dental medicine journals between 1993-2018.PLoS One. 2023 Dec 14;18(12):e0295864. doi: 10.1371/journal.pone.0295864. eCollection 2023. PLoS One. 2023. PMID: 38096136 Free PMC article.
-
A PRISMA assessment of the reporting quality of systematic reviews of nursing published in the Cochrane Library and paper-based journals.Medicine (Baltimore). 2019 Dec;98(49):e18099. doi: 10.1097/MD.0000000000018099. Medicine (Baltimore). 2019. PMID: 31804319 Free PMC article.
-
Partially systematic thoughts on the history of systematic reviews.Syst Rev. 2018 Oct 27;7(1):176. doi: 10.1186/s13643-018-0833-3. Syst Rev. 2018. PMID: 30368251 Free PMC article.
-
Reporting preclinical anesthesia study (REPEAT): Evaluating the quality of reporting in the preclinical anesthesiology literature.PLoS One. 2019 May 23;14(5):e0215221. doi: 10.1371/journal.pone.0215221. eCollection 2019. PLoS One. 2019. PMID: 31120888 Free PMC article. Review.
-
Reporting guidelines for health research: protocol for a cross-sectional analysis of the EQUATOR Network Library.BMJ Open. 2019 Mar 4;9(3):e022769. doi: 10.1136/bmjopen-2018-022769. BMJ Open. 2019. PMID: 30837245 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials