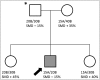
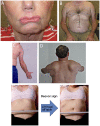
Facioscapulohumeral Muscular Dystrophy
- PMID: 27922500
- PMCID: PMC5898965
- DOI: 10.1212/CON.0000000000000399
Facioscapulohumeral Muscular Dystrophy
Abstract
Purpose of review: This article describes the clinical characteristics, diagnosis, molecular pathogenesis, and treatment of facioscapulohumeral muscular dystrophy (FSHD).
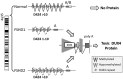
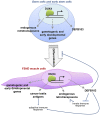
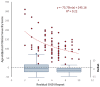
Recent findings: FSHD comprises two genetically distinct types that converge on a common downstream pathway of the expression of the toxic protein DUX4. Approximately 95% of patients have FSHD type 1 (FSHD1), in which loss of DNA repetitive elements (D4Z4 repeats) in the subtelomeric region of chromosome 4q causes decreased methylation and epigenetic derepression of DUX4, a gene contained within each D4Z4 repeat. FSHD type 2 (FSHD2) occurs through a deletion-independent mechanism but, similar to FSHD1, leads to decreased methylation and epigenetic derepression in the same region of chromosome 4q. Whereas FSHD1 is dominantly inherited, FSHD2 shows digenic inheritance, and about 80% of patients will have a mutation in the SMCHD1 gene. DUX4 lacks a polyadenylation signal, so both FSHD1 and FSHD2 only occur in the presence of permissive 4q polymorphisms, which provide a stabilizing polyadenylation sequence. FSHD is an epigenetic disease, and penetrance and severity are related to both the number of residual D4Z4 units and D4Z4 methylation.
Summary: Recent consensus guidelines outline standards for care for FSHD, and identification of potential therapeutic targets have shifted emphasis in the research community toward drug development and clinical trial planning.
Figures

Similar articles
-
[Facioscapulohumeral muscular dystrophy type 2].Rev Neurol (Paris). 2013 Aug-Sep;169(8-9):564-72. doi: 10.1016/j.neurol.2013.02.004. Epub 2013 Aug 20. Rev Neurol (Paris). 2013. PMID: 23969240 French.
-
Genetic and epigenetic characteristics of FSHD-associated 4q and 10q D4Z4 that are distinct from non-4q/10q D4Z4 homologs.Hum Mutat. 2014 Aug;35(8):998-1010. doi: 10.1002/humu.22593. Epub 2014 Jun 24. Hum Mutat. 2014. PMID: 24838473 Free PMC article.
-
A complex interplay of genetic and epigenetic events leads to abnormal expression of the DUX4 gene in facioscapulohumeral muscular dystrophy.Neuromuscul Disord. 2016 Dec;26(12):844-852. doi: 10.1016/j.nmd.2016.09.015. Epub 2016 Sep 19. Neuromuscul Disord. 2016. PMID: 27816329 Review.
-
Identifying diagnostic DNA methylation profiles for facioscapulohumeral muscular dystrophy in blood and saliva using bisulfite sequencing.Clin Epigenetics. 2014 Oct 29;6(1):23. doi: 10.1186/1868-7083-6-23. eCollection 2014. Clin Epigenetics. 2014. PMID: 25400706 Free PMC article.
-
Facioscapulohumeral muscular dystrophy.Neurol Clin. 2014 Aug;32(3):721-8, ix. doi: 10.1016/j.ncl.2014.04.003. Epub 2014 May 15. Neurol Clin. 2014. PMID: 25037087 Free PMC article. Review.
Cited by
-
The variability of SMCHD1 gene in FSHD patients: evidence of new mutations.Hum Mol Genet. 2019 Dec 1;28(23):3912-3920. doi: 10.1093/hmg/ddz239. Hum Mol Genet. 2019. PMID: 31600781 Free PMC article.
-
A female case report of LGMD2B with compound heterozygous mutations of the DYSF gene and asymptomatic mutation of the X-linked DMD gene.Front Neurol. 2023 Sep 27;14:1213090. doi: 10.3389/fneur.2023.1213090. eCollection 2023. Front Neurol. 2023. PMID: 37830096 Free PMC article.
-
Designed U7 snRNAs inhibit DUX4 expression and improve FSHD-associated outcomes in DUX4 overexpressing cells and FSHD patient myotubes.Mol Ther Nucleic Acids. 2020 Dec 10;23:476-486. doi: 10.1016/j.omtn.2020.12.004. eCollection 2021 Mar 5. Mol Ther Nucleic Acids. 2020. PMID: 33510937 Free PMC article.
-
A Promising Future for Stem-Cell-Based Therapies in Muscular Dystrophies-In Vitro and In Vivo Treatments to Boost Cellular Engraftment.Int J Mol Sci. 2019 Oct 31;20(21):5433. doi: 10.3390/ijms20215433. Int J Mol Sci. 2019. PMID: 31683627 Free PMC article. Review.
-
Facio-scapulo-humeral muscular dystrophy with early joint contractures and rigid spine.Acta Myol. 2019 Mar 1;38(1):25-28. eCollection 2019 Mar. Acta Myol. 2019. PMID: 31309179 Free PMC article.
References
-
- Tawil R, Kissel JT, Heatwole C, et al. Evidence-based guideline summary: evaluation, diagnosis, and management of facioscapulohumeral muscular dystrophy: report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology and the Practice Issues Review Panel of the American Association of Neuromuscular & Electrodiagnostic Medicine. Neurology 2015;85(4):357–364. doi:10.1212/WNL.0000000000001783. - PMC - PubMed
-
- Deenen JCW, Horlings CGC, Verschuuren JJGM, et al. The epidemiology of neuromuscular disorders: a comprehensive overview of the literature. J Neuromuscul Dis 2015;2:73–85. doi:10.3233/JND-140045. - PubMed
-
- Statland JM, Tawil R. Risk of functional impairment in facioscapulohumeral muscular dystrophy. Muscle Nerve 2014;49(4):520–527. doi:10.1002/mus.23949. - PubMed
Publication types
MeSH terms
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

