Design Characteristics Influence Performance of Clinical Prediction Rules in Validation: A Meta-Epidemiological Study
- PMID: 26730980
- PMCID: PMC4701404
- DOI: 10.1371/journal.pone.0145779
Design Characteristics Influence Performance of Clinical Prediction Rules in Validation: A Meta-Epidemiological Study
Abstract
Background: Many new clinical prediction rules are derived and validated. But the design and reporting quality of clinical prediction research has been less than optimal. We aimed to assess whether design characteristics of validation studies were associated with the overestimation of clinical prediction rules' performance. We also aimed to evaluate whether validation studies clearly reported important methodological characteristics.
Methods: Electronic databases were searched for systematic reviews of clinical prediction rule studies published between 2006 and 2010. Data were extracted from the eligible validation studies included in the systematic reviews. A meta-analytic meta-epidemiological approach was used to assess the influence of design characteristics on predictive performance. From each validation study, it was assessed whether 7 design and 7 reporting characteristics were properly described.
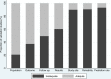
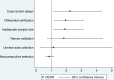
Results: A total of 287 validation studies of clinical prediction rule were collected from 15 systematic reviews (31 meta-analyses). Validation studies using case-control design produced a summary diagnostic odds ratio (DOR) 2.2 times (95% CI: 1.2-4.3) larger than validation studies using cohort design and unclear design. When differential verification was used, the summary DOR was overestimated by twofold (95% CI: 1.2 -3.1) compared to complete, partial and unclear verification. The summary RDOR of validation studies with inadequate sample size was 1.9 (95% CI: 1.2 -3.1) compared to studies with adequate sample size. Study site, reliability, and clinical prediction rule was adequately described in 10.1%, 9.4%, and 7.0% of validation studies respectively.
Conclusion: Validation studies with design shortcomings may overestimate the performance of clinical prediction rules. The quality of reporting among studies validating clinical prediction rules needs to be improved.
Conflict of interest statement
Figures

Similar articles
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
A meta-epidemiological study to examine the association between bias and treatment effects in neonatal trials.Evid Based Child Health. 2014 Dec;9(4):1052-9. doi: 10.1002/ebch.1985. Evid Based Child Health. 2014. PMID: 25504975
-
Reporting and Methodological Quality of Systematic Reviews and Meta-Analyses of Nursing Interventions in Patients With Alzheimer's Disease: General Implications of the Findings.J Nurs Scholarsh. 2019 May;51(3):308-316. doi: 10.1111/jnu.12462. Epub 2019 Feb 25. J Nurs Scholarsh. 2019. PMID: 30806019
-
Does this child have bacterial meningitis? A systematic review of clinical prediction rules for children with suspected bacterial meningitis.J Emerg Med. 2013 Oct;45(4):508-19. doi: 10.1016/j.jemermed.2013.03.042. Epub 2013 Aug 2. J Emerg Med. 2013. PMID: 23910166 Review.
-
Diagnostic management strategies for adults and children with minor head injury: a systematic review and an economic evaluation.Health Technol Assess. 2011 Aug;15(27):1-202. doi: 10.3310/hta15270. Health Technol Assess. 2011. PMID: 21806873 Free PMC article. Review.
Cited by
-
Cardiac Delirium Index for Predicting the Occurrence of Postoperative Delirium in Adult Patients After Coronary Artery Bypass Grafting.Clin Interv Aging. 2021 Mar 17;16:487-495. doi: 10.2147/CIA.S302526. eCollection 2021. Clin Interv Aging. 2021. PMID: 33762820 Free PMC article.
-
Empirical evidence of the impact of study characteristics on the performance of prediction models: a meta-epidemiological study.BMJ Open. 2019 Apr 1;9(4):e026160. doi: 10.1136/bmjopen-2018-026160. BMJ Open. 2019. PMID: 30940759 Free PMC article. Review.
-
Oropouche virus infection in patients with acute febrile syndrome: Is a predictive model based solely on signs and symptoms useful?PLoS One. 2022 Jul 26;17(7):e0270294. doi: 10.1371/journal.pone.0270294. eCollection 2022. PLoS One. 2022. PMID: 35881626 Free PMC article.
-
A framework for meta-analysis of prediction model studies with binary and time-to-event outcomes.Stat Methods Med Res. 2019 Sep;28(9):2768-2786. doi: 10.1177/0962280218785504. Epub 2018 Jul 23. Stat Methods Med Res. 2019. PMID: 30032705 Free PMC article.
-
Predictors for independent external validation of cardiovascular risk clinical prediction rules: Cox proportional hazards regression analyses.Diagn Progn Res. 2018 Feb 6;2:3. doi: 10.1186/s41512-018-0025-6. eCollection 2018. Diagn Progn Res. 2018. PMID: 31093553 Free PMC article.
References
-
- Laupacis A, Sekar N, Stiell IG. Clinical prediction rules. A review and suggested modifications of methodological standards. JAMA: the journal of the American Medical Association. 1997;277(6):488–94. Epub 1997/02/12. . - PubMed
-
- McGinn TG, Guyatt GH, Wyer PC, Naylor CD, Stiell IG, Richardson WS. Users' guides to the medical literature: XXII: how to use articles about clinical decision rules. Evidence-Based Medicine Working Group. JAMA: the journal of the American Medical Association. 2000;284(1):79–84. Epub 2000/06/29. . - PubMed
-
- Justice AC, Covinsky KE, Berlin JA. Assessing the generalizability of prognostic information. Annals of internal medicine. 1999;130(6):515–24. . - PubMed
-
- Reilly BM, Evans AT. Translating clinical research into clinical practice: impact of using prediction rules to make decisions. Annals of internal medicine. 2006;144(3):201–9. Epub 2006/02/08. . - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

