Timing, rates and spectra of human germline mutation
- PMID: 26656846
- PMCID: PMC4731925
- DOI: 10.1038/ng.3469
Timing, rates and spectra of human germline mutation
Abstract
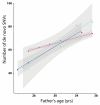
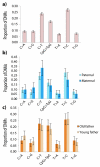
Germline mutations are a driving force behind genome evolution and genetic disease. We investigated genome-wide mutation rates and spectra in multi-sibling families. The mutation rate increased with paternal age in all families, but the number of additional mutations per year differed by more than twofold between families. Meta-analysis of 6,570 mutations showed that germline methylation influences mutation rates. In contrast to somatic mutations, we found remarkable consistency in germline mutation spectra between the sexes and at different paternal ages. In parental germ line, 3.8% of mutations were mosaic, resulting in 1.3% of mutations being shared by siblings. The number of these shared mutations varied significantly between families. Our data suggest that the mutation rate per cell division is higher during both early embryogenesis and differentiation of primordial germ cells but is reduced substantially during post-pubertal spermatogenesis. These findings have important consequences for the recurrence risks of disorders caused by de novo mutations.
Figures



Similar articles
-
Large, three-generation human families reveal post-zygotic mosaicism and variability in germline mutation accumulation.Elife. 2019 Sep 24;8:e46922. doi: 10.7554/eLife.46922. Elife. 2019. PMID: 31549960 Free PMC article.
-
Similarities and differences in patterns of germline mutation between mice and humans.Nat Commun. 2019 Sep 6;10(1):4053. doi: 10.1038/s41467-019-12023-w. Nat Commun. 2019. PMID: 31492841 Free PMC article.
-
Parental somatic mosaicism is underrecognized and influences recurrence risk of genomic disorders.Am J Hum Genet. 2014 Aug 7;95(2):173-82. doi: 10.1016/j.ajhg.2014.07.003. Epub 2014 Jul 31. Am J Hum Genet. 2014. PMID: 25087610 Free PMC article.
-
The developmental basis for germline mosaicism in mouse and Drosophila melanogaster.Genetica. 1998;102-103(1-6):421-43. Genetica. 1998. PMID: 9720293 Review.
-
[Sperm Mosaic Variants and Their Influence on the Offspring].Sichuan Da Xue Xue Bao Yi Xue Ban. 2024 May 20;55(3):535-541. doi: 10.12182/20240560507. Sichuan Da Xue Xue Bao Yi Xue Ban. 2024. PMID: 38948294 Free PMC article. Review. Chinese.
Cited by
-
Somatic mutation landscapes at single-molecule resolution.Nature. 2021 May;593(7859):405-410. doi: 10.1038/s41586-021-03477-4. Epub 2021 Apr 28. Nature. 2021. PMID: 33911282
-
Examining the Effects of Hibernation on Germline Mutation Rates in Grizzly Bears.Genome Biol Evol. 2022 Oct 7;14(10):evac148. doi: 10.1093/gbe/evac148. Genome Biol Evol. 2022. PMID: 36173788 Free PMC article.
-
Large, three-generation human families reveal post-zygotic mosaicism and variability in germline mutation accumulation.Elife. 2019 Sep 24;8:e46922. doi: 10.7554/eLife.46922. Elife. 2019. PMID: 31549960 Free PMC article.
-
Similarities and differences in patterns of germline mutation between mice and humans.Nat Commun. 2019 Sep 6;10(1):4053. doi: 10.1038/s41467-019-12023-w. Nat Commun. 2019. PMID: 31492841 Free PMC article.
-
Exome sequencing of hepatocellular carcinoma in lemurs identifies potential cancer drivers: A pilot study.Evol Med Public Health. 2022 Apr 29;10(1):221-230. doi: 10.1093/emph/eoac016. eCollection 2022. Evol Med Public Health. 2022. PMID: 35557512 Free PMC article.
References
-
- Lindahl T, Wood RD. Quality control by DNA repair. Science. 1999;286:1897–905. - PubMed
-
- Hoeijmakers JH. Genome maintenance mechanisms for preventing cancer. Nature. 2001;411:366–74. - PubMed
-
- Scally A, Durbin R. Revising the human mutation rate: implications for understanding human evolution. Nat Rev Genet. 2012;13:745–53. - PubMed
OnlineMethods-References
-
- Koressaar T, Remm M. Enhancements and modifications of primer design program Primer3. Bioinformatics. 2007;23:1289–1291. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

