Clinical research quo vadis? Trends in reporting of clinical trials and observational study designs over two decades
- PMID: 25883705
- PMCID: PMC4394915
- DOI: 10.14740/jocmr2115w
Clinical research quo vadis? Trends in reporting of clinical trials and observational study designs over two decades
Abstract
Background: Multiple classifications have been developed that classify the medical literature into different levels of evidence to facilitate the evaluation of study results and practice of evidence-based medicine. The suggested hierarchies of evidence are generally based on the type of study design; randomized, controlled clinical trials constitute the top level of evidence while case reports rank the lowest among epidemiologic study designs. However, little is known about the frequency with which different study designs appear in the medical literature overall. The purpose of this study was to describe trends in the frequency of reports of randomized control trials (RCTs) as compared to other study designs in the medical literature over two decades.
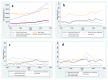
Methods: Data about the prevalence of various types of study designs in the medical literature over the last two decades (years 1990 - 2009) were abstracted from PubMed, validated and subjected to cross-sectional and longitudinal analysis.
Results: In the last 20 years, the annual rate of publication of journal articles has more than doubled. During this period, the percentage of observational studies increased from 29.9% to 40.5%, the percentage of reports of RCTs increased minimally, and there was a striking decline in the percentage of case reports (from 49.8% to 33.6%) in the medical literature overall. In contrast, in three selected, highly cited medical journals, the percentage of reports of RCTs increased by almost 10%. Surprisingly, the percentage of articles classified as case reports also increased (from 36.3% to 43.8%) in these three journals, while the percentage of reports of cohort and case-control studies decreased.
Conclusion: Though the relative frequency of reports from RCTs has not changed substantially in the last 20 years, cohort studies and case-control studies have largely supplanted simple case reports. In contrast, in high impact journals, the representation of RCTs and case reports has increased, with corresponding declines in reports based on other study designs. Further research will be needed to determine whether those trends in publication have resulted in more robust evidence and faster advancement of medical knowledge.
Keywords: Clinical research; Clinical trials; Observational studies; Study design; Trend.
Figures
Similar articles
-
Impact Factors and Prediction of Popular Topics in a Journal.Ultraschall Med. 2016 Aug;37(4):343-5. doi: 10.1055/s-0042-111209. Epub 2016 Aug 4. Ultraschall Med. 2016. PMID: 27490462 English.
-
Most Common Publication Types in Radiology Journals:: What is the Level of Evidence?Acad Radiol. 2016 May;23(5):628-33. doi: 10.1016/j.acra.2016.01.002. Epub 2016 Feb 18. Acad Radiol. 2016. PMID: 26898526
-
Association between pacifier use and breast-feeding, sudden infant death syndrome, infection and dental malocclusion.JBI Libr Syst Rev. 2005;3(6):1-33. doi: 10.11124/01938924-200503060-00001. JBI Libr Syst Rev. 2005. PMID: 27819973
-
Randomized controlled trials and neurosurgery: the ideal fit or should alternative methodologies be considered?J Neurosurg. 2016 Feb;124(2):558-68. doi: 10.3171/2014.12.JNS142465. Epub 2015 Aug 28. J Neurosurg. 2016. PMID: 26315006 Review.
-
Sutureless Aortic Valve Replacement for Treatment of Severe Aortic Stenosis: A Single Technology Assessment of Perceval Sutureless Aortic Valve [Internet].Oslo, Norway: Knowledge Centre for the Health Services at The Norwegian Institute of Public Health (NIPH); 2017 Aug 25. Report from the Norwegian Institute of Public Health No. 2017-01. Oslo, Norway: Knowledge Centre for the Health Services at The Norwegian Institute of Public Health (NIPH); 2017 Aug 25. Report from the Norwegian Institute of Public Health No. 2017-01. PMID: 29553663 Free Books & Documents. Review.
References
-
- Friedman LM, Furberg C, DeMets DL. Fundamentals of clinical trials. 3rd ed. New York: Springer xviii; 1998. p. 361.
LinkOut - more resources
Full Text Sources
Other Literature Sources
