Do surgery journals insist on reporting by CONSORT and PRISMA? A follow-up survey of 'instructions to authors'
- PMID: 25685339
- PMCID: PMC4323756
- DOI: 10.1016/j.amsu.2014.12.003
Do surgery journals insist on reporting by CONSORT and PRISMA? A follow-up survey of 'instructions to authors'
Abstract
Aims: Guidance has been published on how best to report randomised controlled trials (Consolidated Standards of Reporting Trials - CONSORT) and systematic reviews (Preferred Reporting Items for Systematic Reviews and Meta-analysis - PRISMA). In 2011, we reported a low rate of enforcement by surgery journals for submitted manuscripts to conform to these guidelines. The aim of this follow-up study is to establish whether there has been any improvement.
Methods: We studied the 134 surgery journals indexed in the Journal Citation Report. The 'Instructions to Authors' were scrutinised for inclusion of the following guidance: CONSORT, PRISMA, clinical trial registration and systematic review registration.
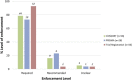
Results: Compared to 2011, there has been an improvement in the endorsement of reporting guidance in journals' 'Instructions to Authors' in 2014, as follows: trial registration (42% vs 33%), CONSORT (42% vs 30%) and PRISMA (19% vs 10%, all p < 0.001). As in 2011, journals with a higher impact were more likely to adopt trial registration (p < 0.001), CONSORT (p < 0.001) and PRISMA (p = 0.002). Journals with editorial offices in the UK were more likely to endorse guidance compared to those outside the UK (p < 0.05). Only one journal mentioned registration for systematic reviews.
Conclusions: Surgery journals are presently more likely to require submitted manuscripts to follow published reporting guidance compared to three years ago. However, overall concordance rates are still low, and an improvement is required to help enhance the quality of reporting - and ultimately the conduct - of randomised control trials and systematic reviews in surgery.
Keywords: Clinical trials; Conflict of interests; Editorial policies; Guidelines; Journalism.
Figures

Similar articles
-
The relationship between endorsing reporting guidelines or trial registration and the impact factor or total citations in surgical journals.PeerJ. 2022 Jan 25;10:e12837. doi: 10.7717/peerj.12837. eCollection 2022. PeerJ. 2022. PMID: 35127293 Free PMC article. Review.
-
Consolidated standards of reporting trials (CONSORT) and the completeness of reporting of randomised controlled trials (RCTs) published in medical journals.Cochrane Database Syst Rev. 2012 Nov 14;11(11):MR000030. doi: 10.1002/14651858.MR000030.pub2. Cochrane Database Syst Rev. 2012. PMID: 23152285 Free PMC article. Review.
-
An Evaluation of Reporting Guidelines and Clinical Trial Registry Requirements Among Orthopaedic Surgery Journals.J Bone Joint Surg Am. 2018 Feb 7;100(3):e15. doi: 10.2106/JBJS.17.00529. J Bone Joint Surg Am. 2018. PMID: 29406351
-
Update on the endorsement of CONSORT by high impact factor journals: a survey of journal "Instructions to Authors" in 2014.Trials. 2016 Jun 24;17(1):301. doi: 10.1186/s13063-016-1408-z. Trials. 2016. PMID: 27343072 Free PMC article.
-
Are pediatric Open Access journals promoting good publication practice? An analysis of author instructions.BMC Pediatr. 2011 Apr 9;11:27. doi: 10.1186/1471-2431-11-27. BMC Pediatr. 2011. PMID: 21477335 Free PMC article.
Cited by
-
Breaking barriers: Navigating the path to successful scientific research publication among faculty members in Egypt.SAGE Open Med. 2024 Sep 17;12:20503121241274710. doi: 10.1177/20503121241274710. eCollection 2024. SAGE Open Med. 2024. PMID: 39296882 Free PMC article.
-
Epidemiology and Reporting Characteristics of Systematic Reviews of Biomedical Research: A Cross-Sectional Study.PLoS Med. 2016 May 24;13(5):e1002028. doi: 10.1371/journal.pmed.1002028. eCollection 2016 May. PLoS Med. 2016. PMID: 27218655 Free PMC article.
-
A Reporting Quality Assessment of Systematic Reviews and Meta-Analyses in Sports Physical Therapy: A Review of Reviews.Healthcare (Basel). 2021 Oct 14;9(10):1368. doi: 10.3390/healthcare9101368. Healthcare (Basel). 2021. PMID: 34683046 Free PMC article. Review.
-
High impact nutrition and dietetics journals' use of publication procedures to increase research transparency.Res Integr Peer Rev. 2020 Aug 31;5:12. doi: 10.1186/s41073-020-00098-9. eCollection 2020. Res Integr Peer Rev. 2020. PMID: 32884841 Free PMC article.
-
The relationship between endorsing reporting guidelines or trial registration and the impact factor or total citations in surgical journals.PeerJ. 2022 Jan 25;10:e12837. doi: 10.7717/peerj.12837. eCollection 2022. PeerJ. 2022. PMID: 35127293 Free PMC article. Review.
References
-
- De Angelis C., Drazen J.M., Frizelle F.A., Haug C., Hoey J., Horton R. Clinical trial registration: a statement from the international committee of medical journal editors. Ann Intern Med. 2004;141(6):477–478. - PubMed
-
- Begg C., Cho M., Eastwood S., Horton R., Moher D., Olkin I. Improving the quality of reporting of randomized controlled trials. The CONSORT statement. JAMA J Am Med Assoc. 1996;276(8):637–639. - PubMed
-
- Plint A.C., Moher D., Morrison A., Schulz K., Altman D.G., Hill C. Does the CONSORT checklist improve the quality of reports of randomised controlled trials? A systematic review. Med J Aust. 2006;185(5):263–267. - PubMed
-
- Moher D., Cook D.J., Eastwood S., Olkin I., Rennie D., Stroup D.F. Improving the quality of reports of meta-analyses of randomised controlled trials: the QUOROM statement. Quality of reporting of meta-analyses. Lancet. 1999;354(9193):1896–1900. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

