Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. The TRIPOD Group
- PMID: 25561516
- PMCID: PMC4297220
- DOI: 10.1161/CIRCULATIONAHA.114.014508
Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. The TRIPOD Group
Abstract
Background: Prediction models are developed to aid health care providers in estimating the probability or risk that a specific disease or condition is present (diagnostic models) or that a specific event will occur in the future (prognostic models), to inform their decision making. However, the overwhelming evidence shows that the quality of reporting of prediction model studies is poor. Only with full and clear reporting of information on all aspects of a prediction model can risk of bias and potential usefulness of prediction models be adequately assessed.
Methods: The Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD) Initiative developed a set of recommendations for the reporting of studies developing, validating, or updating a prediction model, whether for diagnostic or prognostic purposes. This article describes how the TRIPOD Statement was developed. An extensive list of items based on a review of the literature was created, which was reduced after a Web-based survey and revised during a 3-day meeting in June 2011 with methodologists, health care professionals, and journal editors. The list was refined during several meetings of the steering group and in e-mail discussions with the wider group of TRIPOD contributors.
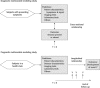
Results: The resulting TRIPOD Statement is a checklist of 22 items, deemed essential for transparent reporting of a prediction model study. The TRIPOD Statement aims to improve the transparency of the reporting of a prediction model study regardless of the study methods used. The TRIPOD Statement is best used in conjunction with the TRIPOD explanation and elaboration document.
Conclusions: To aid the editorial process and readers of prediction model studies, it is recommended that authors include a completed checklist in their submission (also available at www.tripod-statement.org).
Keywords: diagnosis; epidemiology; prognosis; research design; risk; statistics.
© 2015 The Authors.
Figures

Similar articles
-
Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD): The TRIPOD Statement.Eur Urol. 2015 Jun;67(6):1142-1151. doi: 10.1016/j.eururo.2014.11.025. Epub 2015 Jan 5. Eur Urol. 2015. PMID: 25572824
-
Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement.BMJ. 2015 Jan 7;350:g7594. doi: 10.1136/bmj.g7594. BMJ. 2015. PMID: 25569120 Review.
-
Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD Statement.BMC Med. 2015 Jan 6;13:1. doi: 10.1186/s12916-014-0241-z. BMC Med. 2015. PMID: 25563062 Free PMC article.
-
Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement.BJOG. 2015 Feb;122(3):434-43. doi: 10.1111/1471-0528.13244. BJOG. 2015. PMID: 25623578
-
Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis (TRIPOD): the TRIPOD statement.Ann Intern Med. 2015 Jan 6;162(1):55-63. doi: 10.7326/M14-0697. Ann Intern Med. 2015. PMID: 25560714
Cited by
-
A Comprehensive Nomogram Integrating Phonocardiogram and Echocardiogram Features for the Diagnosis of Heart Failure With Preserved Ejection Fraction.Clin Cardiol. 2024 Nov;47(11):e70022. doi: 10.1002/clc.70022. Clin Cardiol. 2024. PMID: 39465895 Free PMC article.
-
Early Detection of In-Patient Deterioration: One Prediction Model Does Not Fit All.Crit Care Explor. 2020 May 11;2(5):e0116. doi: 10.1097/CCE.0000000000000116. eCollection 2020 May. Crit Care Explor. 2020. PMID: 32671347 Free PMC article.
-
Development and validation of a dynamic deep learning algorithm using electrocardiogram to predict dyskalaemias in patients with multiple visits.Eur Heart J Digit Health. 2022 Nov 22;4(1):22-32. doi: 10.1093/ehjdh/ztac072. eCollection 2023 Jan. Eur Heart J Digit Health. 2022. PMID: 36743876 Free PMC article.
-
A methodological checklist for fMRI drug cue reactivity studies: development and expert consensus.Nat Protoc. 2022 Mar;17(3):567-595. doi: 10.1038/s41596-021-00649-4. Epub 2022 Feb 4. Nat Protoc. 2022. PMID: 35121856 Free PMC article.
-
Deep neural networks reveal novel sex-specific electrocardiographic features relevant for mortality risk.Eur Heart J Digit Health. 2022 Mar 21;3(2):245-254. doi: 10.1093/ehjdh/ztac010. eCollection 2022 Jun. Eur Heart J Digit Health. 2022. PMID: 36713005 Free PMC article.
References
-
- Moons KG, Royston P, Vergouwe Y, Grobbee DE, Altman DG. Prognosis and prognostic research: what, why, and how? BMJ. 2009;338:b375. - PubMed
-
- Steyerberg EW. Clinical Prediction Models: A Practical Approach to Development, Validation, and Updating. New York: Springer; 2009.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

